Peptides That Cross Categories
When One Molecule Influences Metabolism, Longevity, Performance, and Repair
Research Use Only Notice: The information presented in this article is for educational and scientific discussion purposes only. All compounds referenced are intended strictly for laboratory research and analytical use. They are not approved for human consumption, not intended to diagnose, treat, cure, or prevent any disease, and are not offered as medical advice. Readers are responsible for compliance with all applicable laws and regulations. Always consult a qualified professional for medical or health-related decisions.
Introduction
Most peptide brands organize research compounds into tidy shelves: Fat Loss, Recovery, Longevity, Cognitive, Performance. It’s clean, simple, and easy to shop. It’s also not how biology works.
Inside living systems, pathways overlap constantly. Metabolism isn’t just “calories in vs calories out”—it’s an endocrine and signaling network that determines what tissues do with fuel. Inflammation isn’t only “swelling”—it’s a messaging layer that can change how cells behave for months or years. Mitochondria aren’t merely “energy factories”—they regulate oxidative stress balance, apoptosis signals, immune triggers, and cellular survival strategies. And aging isn’t a single switch at all; it’s the cumulative consequence of many intertwined stresses, signaling errors, and maintenance failures.
That’s why certain compounds refuse to stay in one category. They touch upstream control nodes—energy sensing, mitochondrial stability, inflammatory tone, vascular function, tissue integrity, circadian regulation. When you influence upstream control nodes, you don’t just get one effect. You often see a cascade.
This is the “category crossover” concept in peptide research: one pathway → multiple domains of function.
Quick Navigation
- The Core Idea
- 1) Retatrutide
- 2) MOTS-c
- 3) SS-31 (Elamipretide)
- 4) Epithalon
- 5) BPC-157
- The Pattern Behind the Pattern
- In Simple Terms
- Peer-Reviewed References
- Compliance Disclaimer
The Core Idea
Why “Cross-Category” Peptides Matter
The most compelling research compounds tend to work upstream of symptoms. Instead of only targeting a single visible outcome, they influence broader physiological conditions—like insulin signaling, mitochondrial efficiency, endothelial function, inflammatory balance, tissue repair dynamics, and cellular stress response. Those conditions shape whether tissues run smoothly or drift toward dysfunction over time.
That’s why “crossover” isn’t just a clever label. It’s a more honest way to describe biology. A molecule doesn’t “belong” to one category. It belongs to the pathways it touches—and pathways rarely sit inside neat marketing boxes.
A helpful framework comes from aging research. The widely cited “Hallmarks of Aging” paper describes core biological processes tied to age-related decline (such as deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, and altered intercellular communication). The practical insight is that these domains are interconnected—when one shifts, others often move with it. That interconnection is exactly why some peptides show relevance across multiple categories. (López-Otín et al., 2013 (Cell; PMC))
Below, we’ll explore several research peptides that commonly “cross categories”—not because they do everything, but because they influence shared control systems that appear in metabolism, performance, recovery, and longevity biology.
1) Retatrutide
Metabolic Modulation + Inflammatory Tone + Longevity-Relevant Signaling
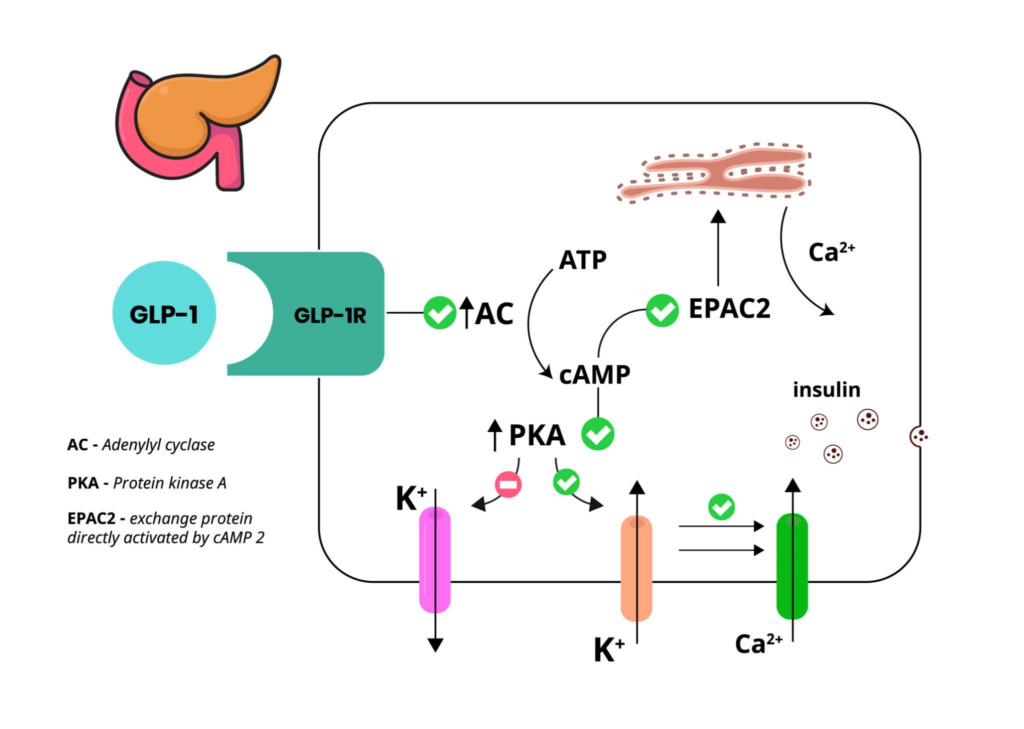
Retatrutide is one of the clearest examples of cross-category biology because its design is explicitly about system-level control. It acts as a triple hormone-receptor agonist—engaging GLP-1, GIP, and glucagon receptors. These are not simple “appetite switches.” They’re part of a coordinated endocrine network that helps govern nutrient intake, nutrient routing, liver glucose output, and whole-body fuel partitioning. In other words, retatrutide is discussed in research not merely as an appetite tool, but as a signal that can shift how the organism handles energy. (Jastreboff et al., 2023 (NEJM))
The crossover begins when metabolism changes the cellular environment. Chronic metabolic dysfunction tends to increase background stress signals: oxidative load, pro-inflammatory signaling, endothelial strain, and disruptions in energy handling. Those are not just “weight” topics—they’re tissue longevity topics, because they influence how quickly cells accumulate damage and how resilient tissues remain under stress. This is why metabolic research repeatedly overlaps with longevity discussions: metabolic stability is upstream of many chronic stress pathways.
A key nuance: this is not a claim that retatrutide is a “longevity drug.” The point is that compounds studied for metabolic endpoints can also intersect longevity-relevant pathways when they reduce upstream pressures that accelerate dysfunction. That intersection is model-dependent and context-dependent, but the biological logic is straightforward.
Retatrutide is commonly discussed in relation to:
- Appetite signaling
- Insulin sensitivity
- Energy expenditure
- Hepatic glucose regulation
Where the crossover begins
Metabolic dysfunction behaves like an accelerant. When insulin signaling is chronically strained, tissues can become less responsive and the body may compensate in ways that amplify systemic stress. Over time, this can correlate with higher oxidative stress, noisier inflammatory messaging, and impaired vascular performance—especially in tissues that live on tight energy margins (endothelium, brain, heart). Those domains are deeply intertwined with aging biology, where mitochondrial stress, altered cell-to-cell communication, and pro-senescent signaling are recurring themes.
This is the “bridge” concept: if a compound improves metabolic efficiency, it may shift the downstream conditions that influence long-term tissue resilience. Even when the primary endpoints are weight and metabolic markers, the same upstream pathways can be connected to broader biological aging patterns.
In Simple Terms
Cleaner metabolism usually means less background cellular stress. Less stress often correlates with lower inflammatory signaling and more stable energy handling. That’s why a compound studied for metabolic outcomes can still overlap with longevity conversations—the systems are biologically connected.
Reta (GLP-3) 20mg
Retatrutide is an investigational multi-agonist peptide engineered to activate three metabolic hormone receptors—GLP-1, GIP, and glucagon—positioning it as a next-generation “triple agonist.” In research settings, it has been examined for its potential influence on appetite regulation, energy utilization, and body-weight–related endpoints. Chemically, retatrutide is a long-acting incretin-mimetic designed to co-activate GLP-1R, GIPR, and GCGR, enabling coordinated engagement of complementary metabolic pathways. Studies have explored its combined effects on glucose homeostasis, gastric emptying, insulin dynamics, lipolysis, and energy expenditure within controlled…
2) MOTS-c
Mitochondrial Signaling + Metabolic Resilience + Performance and Healthspan Models
MOTS-c sits in a particularly interesting crossover zone because it is a mitochondrial-derived peptide encoded within mitochondrial DNA. That alone is conceptually important: it suggests mitochondria are not only energy producers, but also signaling hubs that can send “status updates” to the cell and organism. When mitochondrial status changes, signaling changes—and that can alter metabolism, stress responses, and functional capacity.
One foundational study describes MOTS-c as promoting metabolic homeostasis and reducing obesity and insulin resistance in models. What’s notable here isn’t just the metabolic outcome—it’s the implication that mitochondrial-origin signals can influence systemic nutrient handling. (Lee et al., 2015 (Cell Metabolism; PubMed))
The crossover into performance and aging emerges because mitochondria sit at the center of physical function and cellular resilience. If mitochondria are inefficient, the issue isn’t merely “less energy.” Inefficiency can increase reactive oxygen species signaling, elevate stress responses, and reduce the capacity to adapt under load. That means mitochondrial peptides can logically appear in multiple categories: metabolic efficiency, endurance/performance capacity, recovery dynamics, and longevity biology.
A Nature Communications paper describes MOTS-c as an exercise-induced mitochondrial-encoded regulator tied to age-dependent physical decline and muscle homeostasis in models. Whether someone approaches that literature from a metabolism angle or an aging angle, the same upstream truth appears: mitochondrial signaling influences both. (Reynolds et al., 2021 (Nat Communications; PubMed))
MOTS-c is commonly discussed in relation to:
- Metabolic resilience and homeostasis
- Glucose utilization and insulin sensitivity
- Stress adaptation pathways
- Age-associated physical decline models
In Simple Terms
MOTS-c can be viewed as a mitochondrial “signal” that helps shape how the body handles fuel and stress. Better fuel handling supports metabolic outcomes. Better stress adaptation supports performance. And both themes show up in healthier aging patterns—because mitochondria influence nearly every tissue’s ability to stay functional.
MOTS-c 40mg
MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c) is a 16–amino-acid peptide encoded within the mitochondrial 12S rRNA gene region. It belongs to the family of mitochondrial-derived peptides (MDPs) that function as retrograde signals, coordinating communication between mitochondria and the nucleus to influence metabolic and stress-response pathways. Under metabolic stress—such as glucose restriction or elevated oxidative load—MOTS-c has been observed to translocate to the nucleus, where it can modulate gene expression and support adaptive metabolic regulation.
3) SS-31 (Elamipretide)
Mitochondrial Membrane Stability + Oxidative Stress + Vascular and Aging Models
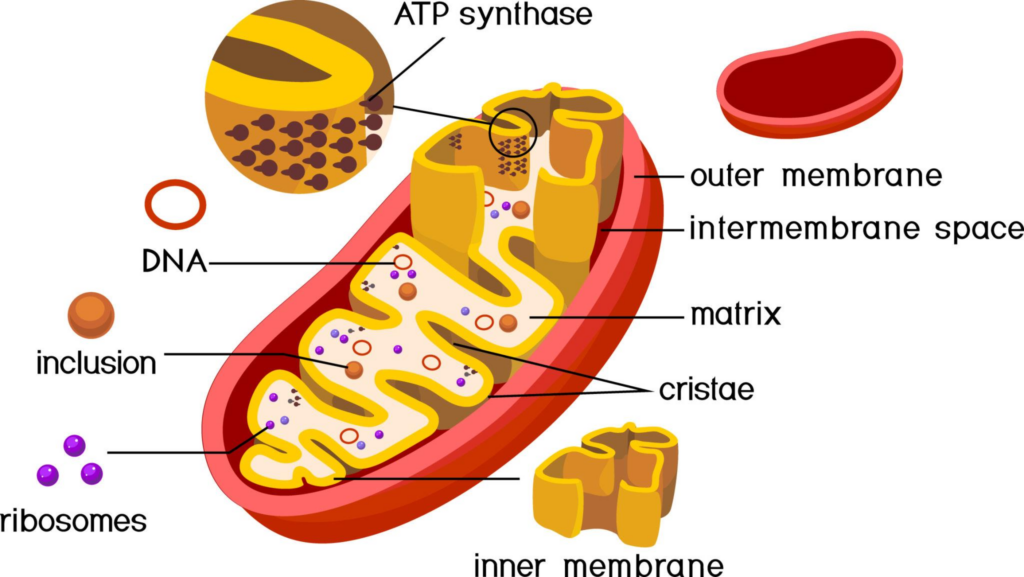
If MOTS-c highlights mitochondrial signaling, SS-31 highlights mitochondrial structure and “clean energy” production. SS-31 (elamipretide) is widely discussed as a mitochondrial-targeted peptide that interacts with cardiolipin-rich regions of the inner mitochondrial membrane. Why does that matter? Because the inner membrane is where electron transport occurs and ATP is generated. When that system becomes inefficient—especially under chronic stress or age-associated decline—reactive oxygen species signaling can rise and overall cellular efficiency can fall.
In a PMC-accessible study in old mice, SS-31 treatment is reported to reverse aspects of age-related cardiac mitochondrial dysfunction, including reductions in mitochondrial ROS and changes in proton leak. The significance isn’t “more energy” as a vague claim; it’s that mitochondrial membrane efficiency and oxidative stress balance are central topics in aging biology. (Chiao et al., 2020 (PMC))
SS-31 also appears in neurovascular aging contexts. Microvascular health helps determine oxygen delivery, nutrient delivery, and waste clearance—factors that matter to brain function over time. An Aging Cell paper reports SS-31 improving neurovascular coupling responses in aged models, illustrating how mitochondrial-targeted interventions can spill into vascular and cognitive-adjacent biology. (Tarantini et al., 2018 (Aging Cell))
This is why SS-31 naturally crosses categories. Mitochondria live in every tissue. Mitochondrial membrane stability influences stress tolerance, oxidative balance, and energy efficiency. Those themes matter to metabolic health, recovery dynamics, vascular function, and aging models—often at the same time.
SS-31 is commonly discussed in relation to:
- Mitochondrial efficiency and oxidative stress balance
- Cardiac aging models
- Neurovascular coupling and microvascular function
- Cellular stress resilience
In Simple Terms
SS-31 isn’t just about “more energy.” It’s about helping mitochondria produce energy more cleanly and with less stress signaling. Cleaner mitochondrial function often means less oxidative strain—a recurring theme in healthier aging and better tissue resilience.
SS-31 40mg
SS-31 (Elamipretide) is a small synthetic peptide designed to target and protect the mitochondria—the “energy centers” of the cell. It binds to a mitochondrial lipid called cardiolipin, helping support healthier energy production, reduce oxidative stress, and maintain mitochondrial stability, especially in high-demand or aging tissues.Because SS-31 works inside the mitochondria rather than on surface receptors, it stands out from most traditional peptides. Research has explored its potential in areas such as energy metabolism, cardiovascular function, neuroprotection, and muscle performance, where…
4) Epithalon (Epitalon)
Cellular Aging Markers + Telomerase-Linked Research + Bioregulation Themes
Epithalon occupies a different crossover lane: cellular aging markers and long-horizon regulation. It’s frequently discussed in relation to telomere biology, including telomerase activity and telomere length in somatic cells. In a paper by Khavinson and colleagues, epithalon is reported to induce telomerase activity and telomere elongation in human somatic cells. Telomere biology is complex and often debated in terms of direct lifespan translation, but the crossover logic is clear: telomere maintenance is part of the language of cellular aging, so anything studied in that context naturally sits in longevity and bioregulation discussions. (Khavinson et al., 2003 (PubMed))
The category overlap expands when you zoom out from cells to systems. Longevity isn’t just a cellular concept—it is also about maintaining stable biological rhythms: repair cycles, immune timing, hormonal signaling, metabolic rhythm. Many “longevity” outcomes are downstream of rhythm and regulation. That’s why compounds discussed in bioregulation contexts often overlap multiple categories, even when the primary language is cellular.
Importantly, “crossover” here doesn’t mean a guarantee of real-world anti-aging outcomes; it means the biology being studied resides at intersections: cellular maintenance pathways and system-level regulation.
Epithalon is commonly discussed in relation to:
- Telomerase activity and telomere length in somatic cells
- Cellular aging markers in experimental settings
- Bioregulation themes related to long-horizon physiology
In Simple Terms
Epithalon shows up in telomere and cellular-aging discussions. And because cellular aging intersects with whole-body regulation (repair cycles, immune rhythm, metabolic stability), it naturally lands in more than one category.
Epithalon 50mg
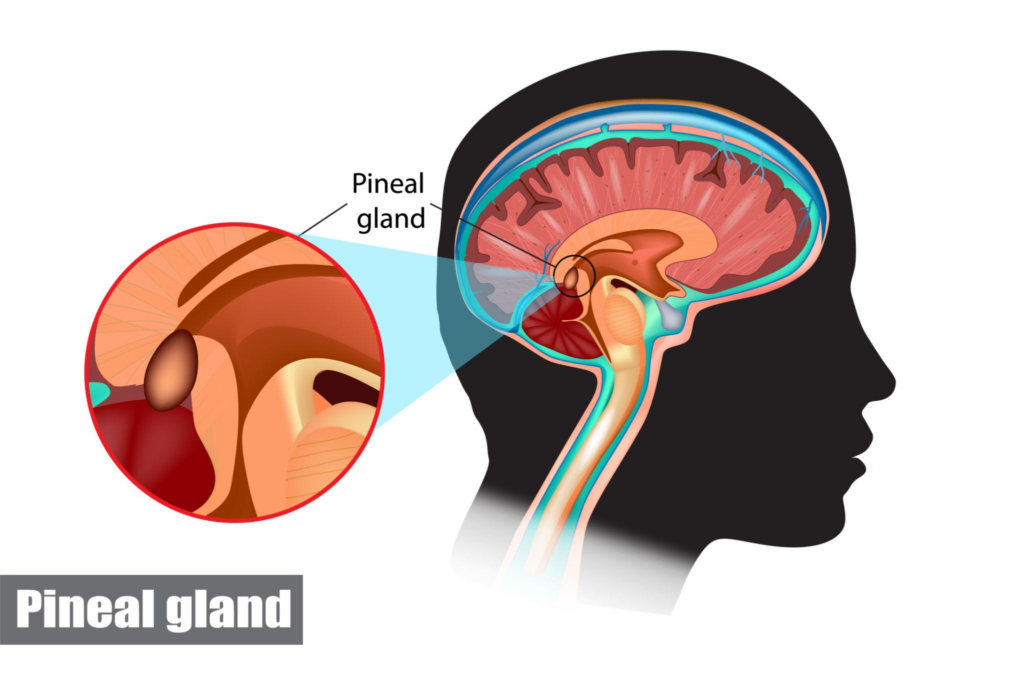
Epithalon, also known as AEDG peptide, Epitalon, or Epithalone, is a synthetic tetrapeptide composed of alanine, glutamic acid, aspartic acid, and glycine. It is modeled after Epithalamin, a naturally occurring pineal gland extract that has been studied for its effects on melatonin production, antioxidant activity, and cellular longevity.Research on Epithalon has suggested potential roles in pineal gland regulation, telomerase activation, oxidative stress reduction, and preservation of retinal and immune system function. Because of its connection to melatonin synthesis and circadian…
5) BPC-157
Tissue Integrity + Gut Barrier Research + Systemic Inflammatory Overlap
BPC-157 is most often associated with recovery and tissue repair research. But the crossover appears when you ask what “recovery” actually means in a systems context. Recovery isn’t only repairing a tendon; it’s restoring tissue integrity while minimizing chronic inflammatory signaling and supporting normal function across the broader environment that tissue lives in—blood flow, immune messaging, and cellular stress response.
Reviews in the scientific literature discuss BPC-157 as a stable gastric pentadecapeptide and provide overviews of observed healing effects across multiple experimental contexts. This is relevant because the GI environment is not isolated: gut barrier integrity can influence systemic inflammation and immune signaling, and systemic inflammation can influence metabolism, vascular function, and long-term tissue resilience. (Sikiric et al., 2011 (PubMed))
Additional discussions (including in Frontiers in Pharmacology) explore wound healing and broader mechanistic themes. Again, the key crossover idea is not “this does everything,” but rather: tissue integrity and inflammatory tone are shared mediators across multiple categories. When inflammatory signaling decreases and tissue repair is supported in models, it can intersect recovery, metabolic stability, and longevity-adjacent resilience themes. (Seiwerth et al., 2021 (Frontiers))
BPC-157 is commonly discussed in relation to:
- Gastrointestinal integrity and protective models
- Tissue repair and wound healing contexts
- Inflammatory signaling overlap in broader reviews
In Simple Terms
BPC-157 gets labeled “recovery,” but recovery is tied to inflammation, gut integrity, and vascular support. Those same systems influence metabolic health and long-term resilience—so it naturally overlaps multiple categories.

BPC-157 10mg
BPC-157, or Body Protection Compound-157, is a synthetic pentadecapeptide derived from a protective protein found in gastric juice. Originally isolated from gastric secretions, this 15-amino acid sequence has been extensively studied in preclinical research for its potential effects on tissue repair, angiogenesis, and cellular protection mechanisms. Research interest centers on its interaction with growth factor pathways and its reported influence on wound healing processes at the molecular level.
The Pattern Behind the Pattern
Why These Compounds Keep Colliding
When you line these peptides up, the crossover becomes less mysterious. Each one is associated with one or more upstream control systems: nutrient sensing and fuel routing, mitochondrial efficiency and oxidative balance, inflammatory signaling and tissue integrity, and cellular maintenance pathways. These domains influence each other, which is why interventions often “echo” across multiple outcomes.
A simple systems-biology view looks like this:
- Nutrient sensing / fuel routing → metabolic stability, inflammatory tone, long-term vascular and tissue stress
- Mitochondrial efficiency → energy output, oxidative stress signaling, stress tolerance, functional capacity
- Inflammatory balance → recovery dynamics, metabolic signaling, endothelial function, long-term resilience
- Cellular maintenance → aging-marker discussions, repair capacity, long-horizon tissue stability
Aging biology frameworks reinforce why this matters: interconnected hallmarks (mitochondrial dysfunction, deregulated nutrient sensing, cellular senescence, altered intercellular communication) feed into each other. That interdependence is exactly why “category crossover” compounds exist at all. (López-Otín et al., 2013 (PMC))
In Simple Terms
Here’s the cleanest way to understand cross-category peptides without getting lost in jargon:
- If a compound improves metabolism, it may also reduce inflammatory stress and improve the cellular environment over time.
- If a compound supports mitochondria, it can influence energy, recovery, vascular function, and aging models—because mitochondria exist in every tissue.
- If a compound supports tissue integrity or gut barrier biology, it may influence systemic inflammation, which spills into metabolism and long-term resilience.
- If a compound is studied in cellular aging markers (like telomere biology), it naturally belongs in longevity discussions—because that’s literally the language of aging research.
The point is not “this peptide does everything.” The point is that these systems are connected—so upstream interventions can show up as multiple downstream outcomes in research models.
Peer-Reviewed References
- Jastreboff AM, et al. Triple–Hormone-Receptor Agonist Retatrutide for Obesity. N Engl J Med. 2023. Link
- Rosenstock J, et al. Retatrutide in type 2 diabetes (randomised phase 2 trial). The Lancet. 2023. Link
- Lee C, et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metabolism. 2015. Link
- Reynolds JC, et al. MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis. Nat Communications. 2021. Link
- Chiao YA, et al. Late-life restoration of mitochondrial function reverses cardiac dysfunction in old mice (SS-31 / elamipretide). PMC Full Text
- Tarantini S, et al. Treatment with the mitochondrial-targeted peptide SS-31 improves neurovascular coupling in aging. Aging Cell. 2018. Link
- Khavinson VK, et al. Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells. Bull Exp Biol Med. 2003. Link
- Sikiric P, et al. Stable gastric pentadecapeptide BPC 157: overview of healing effects. Curr Pharm Des. 2011. Link
- Seiwerth S, et al. Stable gastric pentadecapeptide BPC 157 and wound healing. Frontiers in Pharmacology. 2021. Link
- López-Otín C, et al. The Hallmarks of Aging. Cell. 2013. PMC Full Text

Best selling products
-
Bacteriostatic Water (Reconstitution Solution) 10mL
$16.00 -
Bacteriostatic Water (Reconstitution Solution) 3mL
$9.00 -
Reta (GLP-3) 10mg
$120.00 -
Reta (GLP-3) 20mg
$195.00 -
BPC-157 10mg
$72.00