A foolproof way to reconstitute your lyophilized peptides
Why Getting Peptide Reconstitution Right Matters for Your Research
Disclaimer: The information provided in this article is for educational and informational purposes only. It is not intended as medical advice, diagnosis, or treatment. Products and compounds referenced are for research use only and are not approved for human or veterinary consumption. Always consult a qualified professional regarding health or medical decisions.
Disclaimer: The information provided in this article is for educational and informational purposes only. It is not intended as medical advice, diagnosis, or treatment. Products and compounds referenced are for research use only and are not approved for human or veterinary consumption. Always consult a qualified professional regarding health or medical decisions.

Knowing how to reconstitute lyophilized peptides correctly is one of the most important skills in peptide research — and one of the most commonly mishandled steps. Do it wrong, and you risk degrading your compound before your experiment even begins.
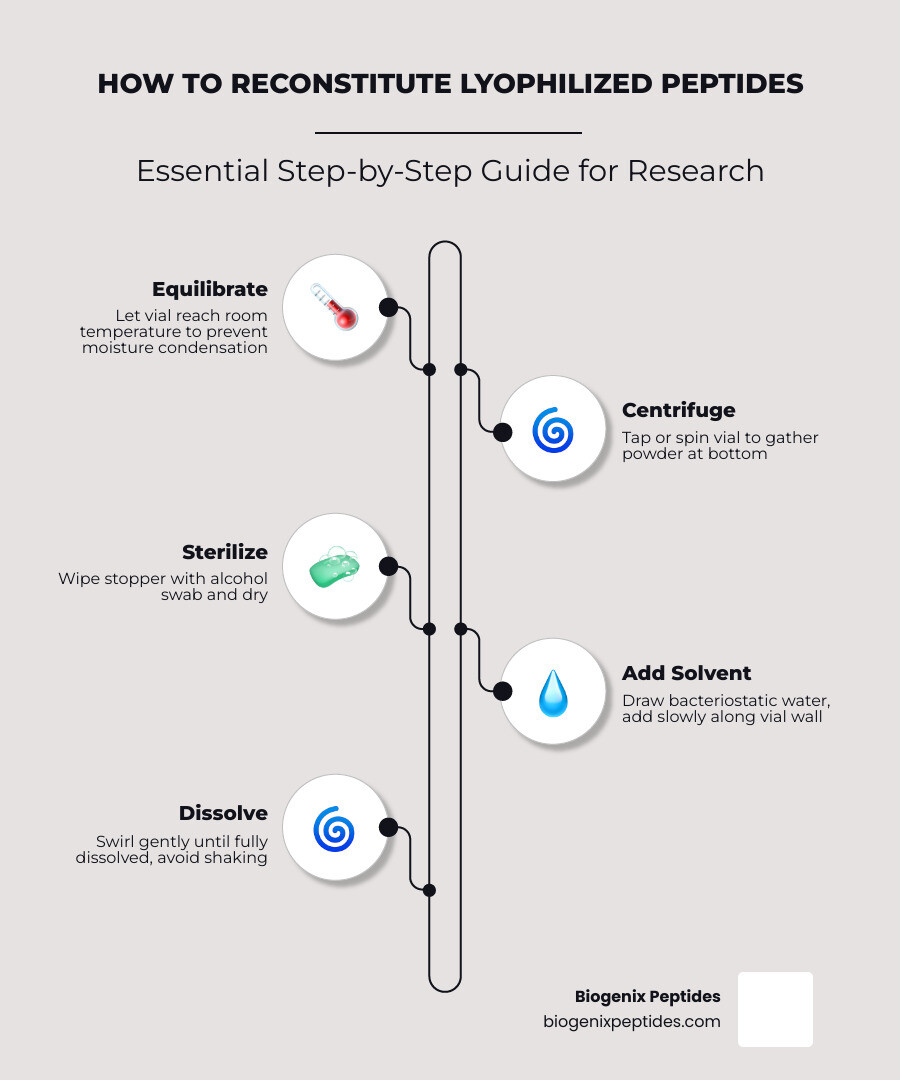
Here is a quick overview of the process:
- Let the vial reach room temperature before opening — prevents moisture condensation on the powder
- Centrifuge or tap the vial to collect all powder at the bottom
- Wipe the stopper with an alcohol swab and let it dry
- Calculate your target volume using: Volume (mL) = Mass (mg) ÷ Desired Concentration (mg/mL)
- Draw your solvent (typically bacteriostatic water) into a sterile syringe
- Add solvent slowly along the vial wall — never spray directly onto the powder
- Swirl gently until fully dissolved — do not shake
- Label and store at 2–8°C, or aliquot for longer-term storage
Lyophilized peptides are freeze-dried compounds preserved in powder form for maximum shelf stability. They can remain stable for years when stored properly — but once reconstituted, that stability window drops significantly. A poorly executed reconstitution can reduce peptide potency by 10–30%, simply from mechanical stress like shaking.
This guide covers everything: solvent selection, step-by-step technique, storage, and troubleshooting — all framed for research purposes only.
I’m Jay Daniel, Founder and CEO of BioGenix Peptides, with years of hands-on experience in peptide science, quality control, and laboratory protocols — including developing best practices for how to reconstitute lyophilized peptides in research settings. In this guide, I’ll walk you through every critical step so your research compounds stay intact and your results stay reliable.
Understanding Lyophilized Peptides and Solvent Selection
Before we pick up a syringe, we need to understand what we are working with. Lyophilization is a sophisticated freeze-drying process that stabilizes peptides by removing water under a vacuum. This process creates a light, porous powder that is highly sensitive to environmental factors. Lyophilization is a freeze-drying process that creates a stable environment for these delicate chains of amino acids, allowing them to be shipped and stored for long periods without losing their structural integrity.
However, lyophilized peptides are hygroscopic. This means they act like tiny sponges, ready to soak up any moisture in the air. If you open a cold vial in a warm room, condensation forms inside immediately. This moisture can partially hydrate the peptide, leading to rapid degradation before you even add your solvent. This is why we always emphasize letting the vial reach room temperature first.
For a deeper dive into why this process is the gold standard, you can read our guide on Why Research Peptides are Typically Lyophilized.
Choosing Between Bacteriostatic and Sterile Water
The most common question we get is about the solvent. For most research applications, you will choose between bacteriostatic water and sterile water.
- Bacteriostatic Water: This is sterile water containing 0.9% benzyl alcohol. The alcohol acts as a preservative, inhibiting the growth of bacteria. When we use bacteriostatic water, the reconstituted solution typically remains stable for 28–30 days when refrigerated at 2–8°C. This makes it the preferred choice for multi-application vials.
- Sterile Water: This is pure water without preservatives. While it is excellent for immediate research use, it lacks antimicrobial protection. Once a vial is breached, any bacteria introduced can multiply quickly. Research suggests that solutions made with sterile water should be used within 3–5 days.
To ensure your research doesn’t go to waste, check out our detailed breakdown: Bacteriostatic Water for Research: Don’t Let Your Research Go Down the Drain. For general solubility rules, Solubility guidelines for peptides can provide additional context on how different amino acid sequences react to various liquids.
Advanced Solvents for Hydrophobic Peptides
Not all peptides love water. Some are “hydrophobic,” meaning they resist dissolving in aqueous solutions. If you add water and the solution remains cloudy or the powder clumps stubbornly, you might need an alternative.
Common advanced solvents include:
- DMSO (Dimethyl Sulfoxide): A powerful solvent that can dissolve almost any peptide. However, it must be used sparingly. Research on DMSO interference with peptide structure shows that high concentrations can disrupt peptide aggregation and structure. We generally recommend using DMSO to create a concentrated stock solution (using less than 10% of the total final volume) and then diluting with your primary buffer.
- Dilute Acetic Acid (0.1%): Often used for basic peptides (those with a positive charge).
- Dilute Ammonia or PBS: Used for acidic peptides to help them reach a stable state.
How to Reconstitute Lyophilized Peptides: A Step-by-Step Guide
Precision is the hallmark of good research. Following a standardized protocol ensures that your results are reproducible and your compounds remain potent.
Preparing the Workspace for How to Reconstitute Lyophilized Peptides
A clean environment is non-negotiable. We recommend working on a non-porous surface that has been sanitized with 70% isopropyl alcohol.
Required Supplies List:
- Your vial of lyophilized peptide
- Reconstitution solvent (e.g., Bacteriostatic Water)
- Sterile syringes (one for mixing, others for aliquoting)
- Alcohol prep pads
- A sharps container for safe disposal (visit safeneedledisposal.org for local guidelines)
- Optional: A centrifuge to pellet the powder
Before starting, ensure you have the Essential Lab Equipment ready. Let the vial sit at room temperature for at least 15–30 minutes. Once ready, pop the plastic cap off the vial to reveal the rubber stopper, and wipe the top of both the peptide vial and the solvent vial with a fresh alcohol swab.
For more details on maintaining a clean field, see our guide on Creating a Sterile Research Environment.
Calculating Concentration for How to Reconstitute Lyophilized Peptides
This is where the math comes in. You need to know the mass of the peptide in the vial (e.g., 5mg or 10mg) and your desired concentration for your research protocol.
The formula is: Volume of Solvent (mL) = Total Peptide Mass (mg) / Target Concentration (mg/mL)
- Example 1: You have a 5mg vial and want a concentration of 2.5mg/mL.
- 5mg / 2.5mg/mL = 2mL. You would add 2mL of bacteriostatic water.
- Example 2: You have a 10mg vial and want a concentration of 5mg/mL.
- 10mg / 5mg/mL = 2mL.
For a deeper look at the 5mg standard, read The Secret to Reconstituting Peptides 5mg Correctly. If you are working with smaller volumes, you might find our Bacteriostatic Water Reconstitution Solution 3ml convenient for precise measurements.
Best Practices for Maintaining Peptide Integrity
Peptides are held together by delicate bonds. If you treat them roughly, those bonds can break—a process called denaturation. Mechanical denaturation can happen much more easily than most researchers realize.
Proper Solvent Addition Techniques
When you are ready to add the solvent, do not simply plunge the liquid into the center of the powder. Most vials are vacuum-sealed, which can cause the solvent to “jet” into the vial, slamming into the lyophilized cake. This high-velocity impact can shear the peptide chains.
Instead, tilt the vial at a 45-degree angle. Aim the needle at the inside glass wall of the vial. Slowly depress the plunger so the liquid trickles down the glass and pools at the bottom, gently hydrating the powder from the base up.
If the vacuum is too strong, you can equalize the pressure by drawing a bit of air into your syringe before inserting it into the vial, or by allowing the vacuum to pull the liquid in slowly while controlling the plunger with your thumb. You can find more on this in our guide: Reconstituting Lyophilized Peptides Step-by-Step 2.
| Action | Impact on Peptide Integrity |
|---|---|
| Gentle Swirling | Maintains 100% potency; allows natural dissolution. |
| Vigorous Shaking | Can reduce potency by 10–30%; causes foaming and denaturation. |
| Direct Spraying | Mechanical stress can shear delicate peptide bonds. |
| Room Temp Equilibration | Prevents moisture-induced degradation. |
Peptide-Specific Reconstitution Tips
Different research compounds have different “personalities” when it comes to dissolving.
- BPC-157: This is a relatively robust, small peptide. It typically dissolves very quickly—often within 30 seconds to 2 minutes of gentle swirling. It is generally very stable in bacteriostatic water.
- Semaglutide: Being a larger, more complex GLP-1 analog, it may take a little longer to fully clear. Some research suggests it can remain stable for up to 56 days when refrigerated, though we recommend the standard 30-day window for maximum reliability.
- TB-500: This peptide is highly sensitive to mechanical stress. Extra care should be taken to avoid any bubbles or foam during the process.
For a comprehensive list of handling protocols, see our Best Practices for Peptide Storage & Handling.
Storage, Stability, and Troubleshooting
Once the peptide is in solution, the clock starts ticking. In its lyophilized state, a peptide might last years in a freezer. Once reconstituted, it is much more vulnerable to heat, light, and bacterial contamination.
Long-Term Stability and Aliquoting
If your research protocol spans several months, do not leave the entire reconstituted vial in the refrigerator. Instead, use “aliquoting.” This involves dividing the solution into several smaller, single-application sterile tubes.
- Avoid Freeze-Thaw Cycles: Every time you freeze and then thaw a liquid peptide, ice crystals form and melt. These crystals act like tiny knives, shredding the peptide structure. This is known as cryodenaturation.
- Proper Freezing: If you must freeze a reconstituted solution, do it once. Aliquot the solution into volumes greater than 20μL to minimize surface-area-to-volume issues, freeze them, and thaw only what you need for that day’s research.
For more on this, refer to Peptide Storage Stability Guidelines.
Identifying Degradation and Solubility Issues
What should your solution look like? In 99% of cases, it should be as clear as bottled water.
- Cloudiness or Opacity: This is a red flag. It usually indicates either that the peptide has precipitated out of the solution or that bacterial contamination has occurred. If a solution becomes cloudy after being clear, it should be discarded.
- Particulates or “Flakes”: Sometimes, small flakes of the lyophilized cake remain. If gentle swirling doesn’t fix it, try placing the vial in the refrigerator for 1–2 hours. Often, the slow hydration at cold temperatures helps these last bits dissolve.
- Color Changes: Any shift in color is a sign of chemical degradation (often oxidation).
If you encounter stubborn solubility issues, you might need a different volume of solvent. Check out our Bacteriostatic Water Reconstitution Solution 10ml if your research requires larger dilution ratios to achieve clarity.
Frequently Asked Questions about Reconstitution
What should the solution look like after mixing?
The final solution should be completely transparent and free of any visible particles, floating “strings,” or cloudiness. Think of it like a vial of pure water. If you see any tint or turbidity, the peptide may not be fully dissolved or may have degraded.
Can I freeze peptides after they have been reconstituted?
While you can freeze them, it is generally not recommended unless you are aliquoting for long-term storage. Repeatedly freezing and thawing a single vial will destroy the peptide’s effectiveness very quickly. If you do freeze, ensure you use a “frost-free” freezer with caution, as temperature fluctuations during the defrost cycle can also cause damage.
How long do reconstituted peptides remain stable in the refrigerator?
When using bacteriostatic water, most peptides are stable for about 30 days. Some researchers find that specific compounds like Semaglutide can last longer, but for the sake of experimental integrity, 30 days is the industry-standard cutoff. After this point, the benzyl alcohol may lose its effectiveness, and the peptide itself may begin to break down into inactive fragments.
Conclusion
Mastering the art of how to reconstitute lyophilized peptides is about more than just mixing powder and water; it’s about preserving the “biological message” encoded in the peptide sequence. By choosing the right solvent, maintaining a sterile environment, and using gentle techniques, you ensure that your research is based on high-quality, potent compounds.
At Biogenix Peptides, we are committed to providing researchers with the highest quality lyophilized products and the knowledge to handle them correctly. For more information on the benefits of our freeze-drying process, visit Why Research Peptides are Typically Lyophilized and How This Benefits Research.
Proper handling is the bridge between a successful experiment and a wasted vial. Take your time, follow the steps, and let the science lead the way.




