What Makes One Peptide 10x More Effective Than Another—Even With the Same Name?
Why purity, stability, and manufacturing quality matter more than most researchers realize.
Two bottles on two different websites. Both are labeled with the same peptide name—BPC-157, CJC-1295, Semaglutide, or Epithalon.
Same molecule. Same name. But somehow, the research outcomes are dramatically different.
Why?
Because peptides are not commodities. A peptide’s effectiveness depends on how it was built, how it was purified, how it was stabilized, and even which carrier molecules or excipients were used.
This is exactly why BioGenix focuses on ultra-high purity, verified U.S. lab testing, and cold-chain–protected stability. But before we talk quality standards, let’s look at the science.
The Science Behind Why Peptides Perform Differently
1. Purity Levels: The Hidden 5–20% That Changes Everything
Scientific View
Peptide synthesis, typically performed via solid-phase peptide synthesis (SPPS), generates not only the target peptide but also a range of by-products, including:
- Truncated peptide chains
- Deletion sequences
- Racemized amino acids
- Oxidized fragments
- Aggregates and misfolded chains
A peptide listed as “≥95% purity” can legally contain up to 5% impurities. If those impurities include truncated analogs that still interact with biological targets, they can alter pharmacodynamics, reduce binding affinity, or create competition at receptor sites.
A peptide purified to 98–99% can deliver several-fold superior functional activity compared to a 90–94% preparation, simply because impurities act as biological noise.
In Simple Terms
Think of peptides like instruction manuals. A high-purity peptide is a clean, complete manual. A lower-purity peptide is the same manual full of missing pages and typos. It might still “kind of” work, but it won’t guide the process as clearly or consistently.
2. Manufacturing Process & Reactor Quality
Scientific View
Peptides synthesized in high-precision, low-contamination reactors minimize:
- Cross-sequence contamination from previous runs
- Residual organic solvents
- Heavy metal contamination
- Excess trifluoroacetate (TFA) or other counterions
- Incorrect stereochemistry at chiral centers
Low-budget reactors and poor process control can yield peptides that match the target sequence on paper but display poor biological activity due to subtle structural defects and contaminants.
In Simple Terms
Cheap labs can technically “make” the peptide, just like cheap factories can technically make engine parts. But when you push them into real-world use, the difference in performance becomes obvious. Better equipment and tighter process control produce better molecules.
3. Peptide Folding & Secondary Structure Integrity
Scientific View
Some peptides rely on specific secondary or tertiary structures for optimal receptor binding, including:
- Correct β-turn formation
- Cyclization or head-to-tail linkage
- Disulfide bridge formation between cysteine residues
- Stable α-helical regions
Even when two preparations share the same amino acid sequence, differences in folding can dramatically alter receptor affinity, biological half-life, and downstream signaling.
In Simple Terms
Imagine two paper airplanes made from the same sheet of paper. One glides across the room; the other nose-dives instantly. The difference is in how they were folded. With peptides, the “folding” of the chain decides how well it works.
4. Carrier Molecules & Excipients: The Silent Performance Factor
Scientific View
Peptide stability and solubility are heavily influenced by excipients such as:
- Mannitol
- Trehalose
- Glycine
- Arginine
- Citrate or phosphate buffers
Properly chosen stabilizers can protect against oxidation, moisture-induced degradation, and aggregation. Poor or incompatible excipients can accelerate breakdown, promote clumping, and reduce bioactivity.
In some cases, optimized excipients can extend peptide stability by an order of magnitude compared to unstabilized formulations.
In Simple Terms
The peptide is the “active ingredient,” but the excipients act like packaging and insulation for the molecule. Good insulation keeps it safe, potent, and easy to work with. Bad insulation lets it spoil before it has a chance to do its job.
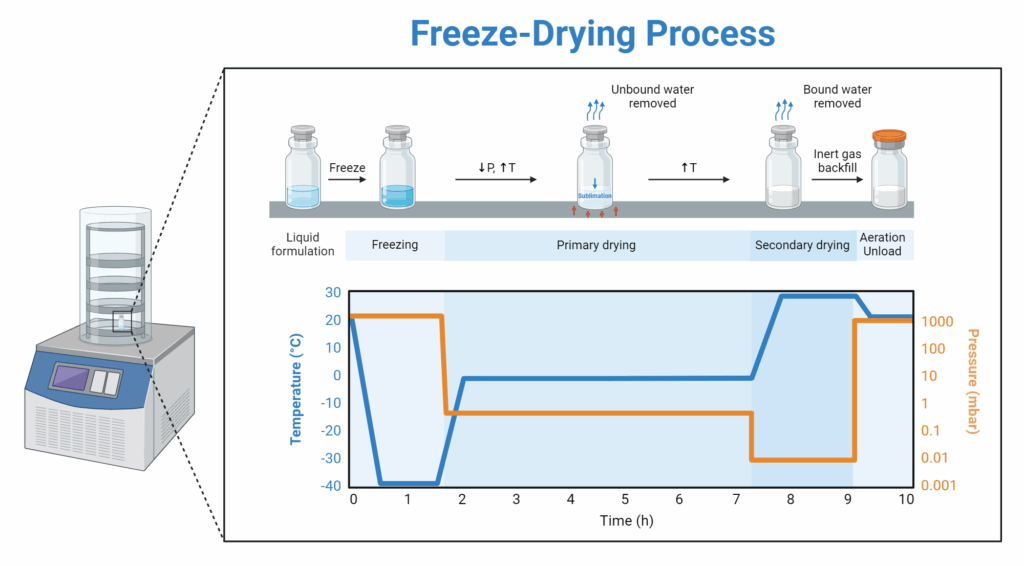
5. Lyophilization Quality (Freeze-Drying)
Lyophilization (Freeze-Drying)
Scientific View
Pharmaceutical-grade lyophilization carefully controls:
- Freezing rate and ice crystal formation
- Primary and secondary drying phases
- Chamber pressure and temperature
- Residual moisture content
Proper lyophilization protects the peptide’s structure and long-term stability. However, cake appearance is not a reliable marker of quality.
Many manufacturers add bulking agents or stabilizers (such as mannitol or glycine) to create a large, fluffy, uniform cake. This looks “perfect” in the vial, but those additives exist primarily for cosmetics and handling—not for research value.
BioGenix does not use unnecessary fillers, binders, or bulking agents. Our peptides are lyophilized in their pure form, so the resulting cake can be firm, thin, fragile, or powdery and may fracture during shipping. This is normal behavior for high-purity peptide mass with no stabilizers holding it together.
In Simple Terms
Some peptides come out of the vial as a solid, even cake. Others look like loose, dusty, or broken powder. That difference is mostly about cosmetics, not quality.
When no bulking agents are added, the actual amount of peptide is very small and naturally brittle. Even gentle movement during shipping can cause the cake to crack or crumble. This is especially common with:
- IGF peptides (IGF-1 LR3, IGF-1 DES)
- HGH Fragment 176-191
- GHRP-2, GHRP-6 and similar GHRPs
- Short bioregulator peptides (Epithalon, Pinealon, Cortagen, etc.)
These peptides have low physical mass and often form very delicate structures when freeze-dried, so they are more likely to look fragmented or powdery in the vial.
The important part: a broken cake does not mean the peptide is damaged or weaker. Once sterile diluent is added, the material dissolves and performs the same whether it started as a firm cake or a loose powder.
BioGenix chooses purity over cosmetic fluff. What you see in the vial is the peptide itself—not a “puffed-up” cake created by fillers.
6. Storage, Transport, & Cold-Chain Integrity
Scientific View
Peptides are sensitive to temperature, light, and moisture. Extended exposure to elevated temperatures can:
- Increase oxidation of sensitive residues (e.g., methionine, tryptophan)
- Promote hydrolysis of peptide bonds
- Encourage aggregation and insolubility
A peptide that experiences prolonged temperature excursions during shipping or storage may lose a significant portion of its functional potency before it is ever used.
In Simple Terms
If a peptide spends a couple of hot days in transit, it is like leaving sushi in a warm car. It might still look okay, but it is definitely not what you were expecting in terms of quality.
7. Why the Lab’s Testing Method Determines Whether You Can Trust the Numbers
Scientific View
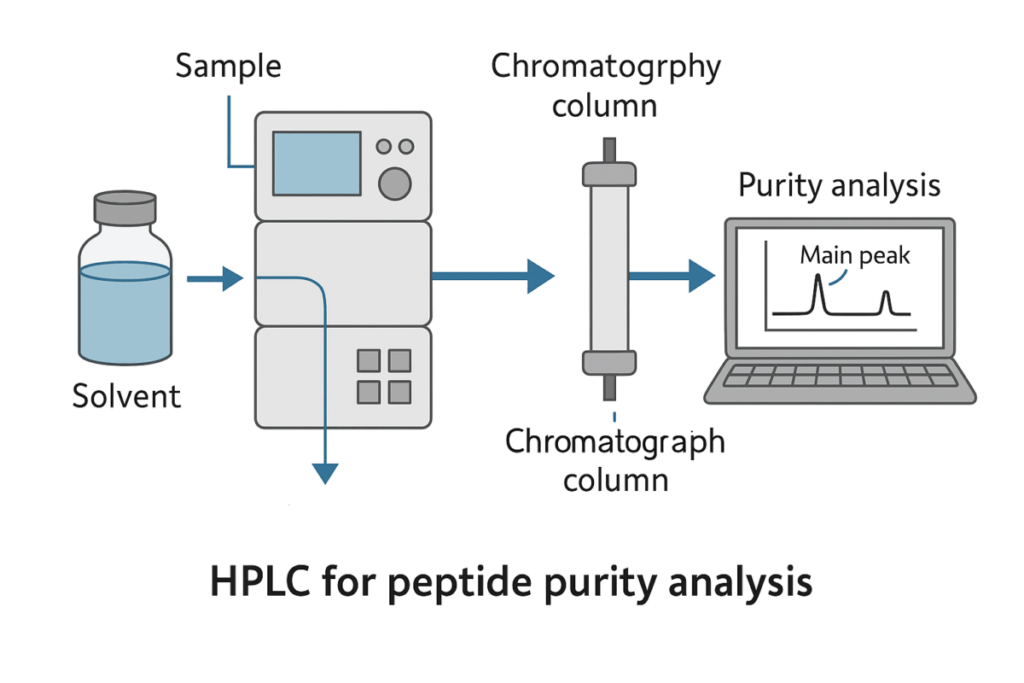
Not all certificates of analysis (COAs) are created equal. Common analytical tools include:
- HPLC (High-Performance Liquid Chromatography) – measures purity and detects impurities.
- Mass Spectrometry (MS) – confirms molecular weight and identity.
- NMR (Nuclear Magnetic Resonance) – can validate structure for complex molecules.
- Endotoxin and microbial testing – assesses cleanliness of production conditions.
Labs that combine HPLC with mass spectrometry provide a much stronger verification of both purity and identity than labs relying on HPLC alone.
In Simple Terms
Some companies only test “how much stuff is in the vial.” Better labs test whether the stuff is actually the right molecule in the first place. That difference matters.
Why Two Identical Peptide Names Can Give You 10x Different Results
Behind the scenes, two products with the same peptide name may be radically different:
- One is made in a certified clean facility; the other in an unregulated lab.
- One is purified to ≥99%; the other to 90–95%.
- One uses pharmaceutical-grade stabilizers; the other uses cheap or inappropriate excipients.
- One is protected by a maintained cold chain; the other experiences multiple heat cycles in transit.
From the outside, they share the same label. In actual research, they behave like completely different products.
The BioGenix Difference
Not hype—chemistry.
BioGenix peptides are produced and tested to meet demanding research expectations, including:
- U.S.-verified laboratory testing
- Ultra-high purity (typically ≥99%, with many lots reaching 99.9%+)
- HPLC and mass spectrometry validation for identity and purity
- Controlled, pharmaceutical-grade lyophilization
- Thoughtfully selected excipients for stability, not dilution
- Cold-chain–aware storage and logistics
- Batch-specific certificates of analysis
- Transparent documentation and ingredient disclosure
This approach is designed to eliminate guesswork and protect the integrity of your research.

Conclusion
Two peptides can share the same name and sequence yet deliver completely different research outcomes. The difference between good results, no results, unexpected side effects, or breakthrough findings often comes down to details most people never see: purity, folding, lyophilization, stabilizers, and the integrity of the cold chain.
When these factors are handled correctly, a peptide becomes predictable, stable, and highly effective. When they are not, you are essentially paying for a label, not a molecule.
By prioritizing analytical transparency, pharmaceutical-style stabilization, and ultra-high purity, BioGenix aims to provide peptides that behave the way serious researchers expect them to.


