The Science of the Bacteriostatic Water Peptide Ratio
Why Getting the Bacteriostatic Water Peptide Ratio Right Matters
Reconstituting lyophilized peptides is the process of dissolving freeze-dried peptide powder into a liquid solvent — most commonly bacteriostatic water — to create a stable, usable solution for research.
If you need a quick answer, here’s the core process at a glance:
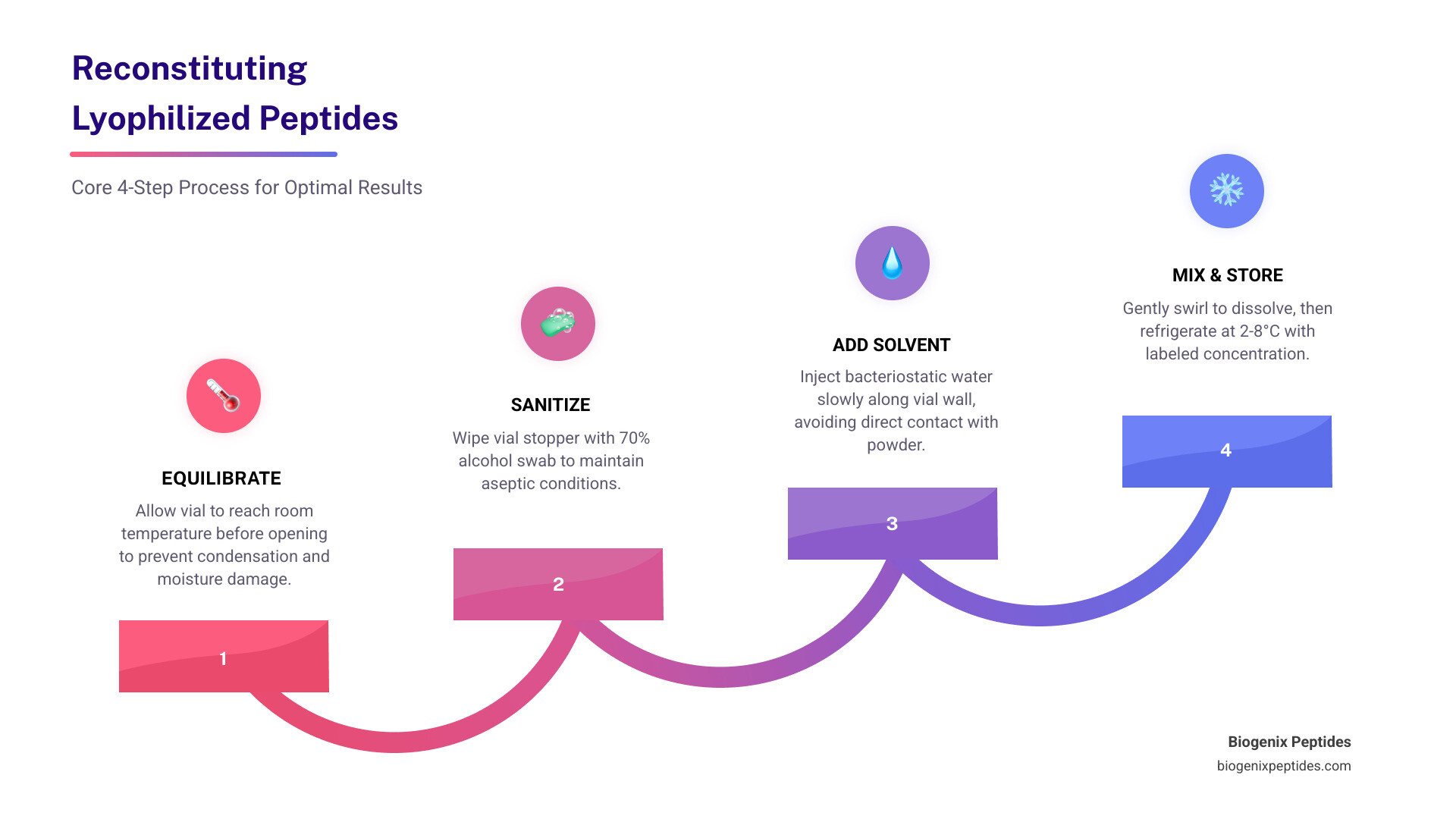
- Equilibrate – Let the vial reach room temperature before opening
- Sanitize – Wipe the vial stopper with a 70% alcohol swab
- Calculate – Determine your solvent volume using: mg/mL = peptide mass ÷ solvent volume
- Add solvent – Inject slowly along the vial wall, never directly onto the powder
- Mix gently – Swirl the vial; never shake or vortex
- Store – Refrigerate at 2-8°C and label with the date and concentration
Getting this right matters more than most researchers realize. The ratio of bacteriostatic water to peptide directly controls your working concentration — and even small errors can throw off every downstream measurement.
Lyophilized peptides are highly hygroscopic, meaning they absorb moisture from the air quickly. They’re also sensitive to mechanical stress during mixing. The wrong solvent volume, the wrong technique, or the wrong storage temperature can degrade a peptide before a single experiment runs.
This guide walks through everything — from solvent selection to dosage math to troubleshooting — so your reconstitution is accurate every time.
I’m Jay Daniel, Founder and CEO of BioGenix Peptides, and through years of hands-on experience in peptide research and quality control, I’ve developed deep expertise in reconstituting lyophilized peptides across a wide range of sequences and applications. I’ll walk you through the science and the practical steps so you can work with confidence in the lab.
Understanding the Process of Reconstituting Lyophilized Peptides
To understand why we spend so much time talking about reconstituting lyophilized peptides, we first have to look at why they come as a powder in the first place. Lyophilization, or freeze-drying, is a sophisticated stabilization process that removes water from a biological sample through sublimation—turning ice directly into vapor under a vacuum.
This process is vital because peptides are inherently “fragile” molecules. In an aqueous (liquid) environment, they are prone to hydrolysis and enzymatic degradation. By removing the water, we essentially put the peptide into a state of suspended animation. According to scientific research on peptide lyophilization, this creates a light, porous “cake” or powder that preserves the structural integrity and biological activity of the molecule for years.
However, this dry state makes the peptide extremely hygroscopic—it wants to suck moisture out of the air the second it gets the chance. This is why research peptides are typically lyophilized; it ensures they remain shelf-stable during shipping and long-term storage. When we reconstitute them, we are carefully reintroducing a solvent to return the peptide to a liquid state where it can interact with receptors or enzymes in a lab setting. If we do this too aggressively or with the wrong pH, we risk “denaturing” the peptide—essentially breaking its specific 3D shape and rendering it useless for research.
Essential Supplies and Solvent Selection: Bacteriostatic vs. Sterile Water
Before you even touch a vial, you need the right gear. Think of this like a high-stakes chemistry bake-off; you wouldn’t use a rusty whisk, and you certainly shouldn’t use tap water.
The Solvent Showdown
The most critical choice you’ll make is the solvent. While “water is water” might seem true, in reconstituting lyophilized peptides, the type of water matters immensely.
- Bacteriostatic Water: This is sterile water containing 0.9% benzyl alcohol. The alcohol acts as a preservative, inhibiting the growth of bacteria. If you plan on using a vial multiple times over several weeks, you should shop bacteriostatic water 10ml or similar sizes. It allows the solution to remain viable for up to 28–30 days when refrigerated.
- Sterile Water for Injection: This is pure, sterile water without any preservatives. It is excellent for single-use applications but lacks the antimicrobial “shield” of bacteriostatic water. If you use sterile water, the solution should typically be used within 3–5 days, as there is nothing to stop bacterial colonies from forming once the stopper has been pierced.
Your Lab Kit
Beyond the solvent, you will need:
- Alcohol Swabs: 70% isopropyl alcohol is the standard for maintaining an aseptic environment.
- Syringes: Typically, a larger “mixing syringe” (3ml) is used to add the solvent, while smaller insulin-style syringes are used for precise dosing. You can find more details in our guide on essential lab equipment for research setups.
- Sharps Container: Safety first! Always follow safe needle disposal guidelines by using a puncture-resistant container.
Protocol for Success: Reconstituting Lyophilized Peptides Safely
Now, let’s get into the “how-to.” Precision is your best friend here.
Step 1: Temperature Equilibration
Never open a cold vial. When you take a vial out of the freezer (-20°C), let it sit on the counter until it reaches room temperature. If you open it while cold, condensation (moisture) will form inside the vial. “Hygroscopic” nature we mentioned? That moisture will instantly begin degrading your peptide.
Step 2: The Aseptic Swipe
Cleanliness is next to… well, successful research. Wipe the top of the peptide vial and the bacteriostatic water vial with a fresh alcohol swab. Let them air dry for 30 seconds. Do not blow on them to speed it up—your breath is full of bacteria! This is the foundation of creating a sterile research environment.
Step 3: Pressure Equalization
Vials are often vacuum-sealed. If you just plunge a needle in, the vacuum might suck the solvent in so fast it “slams” into the powder, causing mechanical damage. To prevent this, draw a volume of air into your syringe equal to the amount of water you plan to add. Inject the air into the water vial first to make drawing the liquid easier, then draw your water. When you move to the peptide vial, insert the needle and let the vacuum gently pull the liquid in, or “burp” the vial by drawing out a bit of air to equalize the pressure.
For a more detailed breakdown, see our reconstituting lyophilized peptides step-by-step guide.
Best Practices for Reconstituting Lyophilized Peptides
When adding the solvent, the “Golden Rule” is: Aim for the wall. Do not spray the water directly onto the powder cake. Instead, let the liquid trickle down the inside glass of the vial. This prevents foaming and “mechanical denaturation”—the physical breaking of peptide bonds due to shear force.
Once the liquid is in, gentle swirling is the only way to go. Imagine you are swirling a very expensive glass of wine. Never shake the vial. Shaking creates air bubbles, and the air-liquid interface can cause the peptides to unfold (denature) or clump together. Following scientific guidelines on peptide solubility ensures that the delicate molecular structure remains intact.
Troubleshooting Issues When Reconstituting Lyophilized Peptides
Sometimes, despite your best efforts, things don’t go perfectly. Here is how to handle the most common hiccups:
- Cloudiness or Particulates: If the solution looks like a snow globe, the peptide hasn’t fully dissolved. This often happens with hydrophobic (water-fearing) peptides. First, try refrigerating the vial for 1-2 hours; sometimes a cold soak helps. If that fails, gentle sonication (sound waves) or a very slight adjustment in pH may be needed.
- Clumping: If the powder turns into a sticky ball, it’s likely a solubility issue. For some basic peptides, adding a drop of 0.1% acetic acid can help. For very stubborn, hydrophobic sequences, researchers sometimes use a tiny amount of DMSO (Dimethyl Sulfoxide), though research on DMSO and peptide aggregation suggests using it sparingly as it can interfere with some biological assays.
- Foaming: If you accidentally shook the vial and it’s full of bubbles, don’t panic. Let it sit in the refrigerator for an hour. The bubbles will eventually settle, and you can assess if the peptide is still in solution.
Avoid these and other pitfalls by reviewing the 10 most common mistakes in peptide research.
Calculating the Bacteriostatic Water Peptide Ratio and Dosage
Math is where the “Science” in our title really lives. You need to know exactly how much peptide is in every drop of liquid. The formula is simple:
Concentration (mg/mL) = Total Peptide Mass (mg) ÷ Total Solvent Volume (mL)
For example, if you have a 5mg vial and you add 2mL of bacteriostatic water, your concentration is 2.5mg/mL.
Comparison Table: Common Dilution Ratios
| Peptide Vial Size | Solvent Added (mL) | Resulting Concentration |
|---|---|---|
| 2 mg | 1 mL | 2 mg/mL |
| 2 mg | 2 mL | 1 mg/mL |
| 5 mg | 1 mL | 5 mg/mL |
| 5 mg | 2 mL | 2.5 mg/mL |
| 5 mg | 5 mL | 1 mg/mL |
| 10 mg | 2 mL | 5 mg/mL |
When calculating, the powder itself has a very small “volume displacement,” but for most research purposes, we focus on the volume of solvent added. For high-precision work, always refer to how to reconstitute lyophilized proteins for advanced math.
Storage and Stability Guidelines for Reconstituted Solutions
Once reconstituting lyophilized peptides is complete, the “stability clock” starts ticking. While lyophilized powder can last 2–4 years in a freezer, the liquid version is much more “perishable.”
The 28-Day Rule
For most peptides reconstituted with bacteriostatic water, 28 days is the standard cutoff for peak potency. After this, the benzyl alcohol begins to lose its effectiveness, and the peptide itself may begin to fragment.
Temperature is King
Always store your reconstituted vials in the refrigerator at 2-8°C (36-46°F). Do not store them in the door of the fridge, as the temperature fluctuates every time you grab a snack. Keep them in the back or in a dedicated drawer.
To Freeze or Not to Freeze?
This is a debated topic. Generally, we recommend aliquoting. If you have a large amount of solution, divide it into small, single-use tubes (aliquots) and freeze those. This way, you only thaw what you need. Repeatedly freezing and thawing a single vial creates “cryodenaturation”—ice crystals physically shredding the peptide chains. Our peptide storage and stability guidelines provide a deeper dive into these environmental factors.
For more on handling, check out best practices for peptide handling.
Frequently Asked Questions
What should I do if the peptide doesn’t dissolve completely?
First, give it time. Some peptides take 15–30 minutes to fully hydrate. If it’s still cloudy, move it to the refrigerator for an hour. If particulates remain, you may need to check the solubility support pages to see if your specific peptide requires a different pH (like a tiny amount of dilute acetic acid for basic peptides).
How long do reconstituted peptides remain stable?
In bacteriostatic water, most remain stable for 28–30 days if refrigerated. Some very stable peptides (like Semaglutide) may last up to 56 days, while others (like IGF-1) are much more fragile and should be used sooner. Signs of degradation include cloudiness, a change in color, or the appearance of “floaties” in the liquid.
Can I freeze peptides after they have been reconstituted?
You can, but it’s risky. If you must freeze them, do it in single-use aliquots to avoid multiple freeze-thaw cycles. Be aware that the formation of ice crystals can cause “cryodenaturation,” which may reduce the biological activity of the peptide.
Conclusion
Mastering the art of reconstituting lyophilized peptides is a fundamental skill for any serious researcher. By understanding the delicate balance of the bacteriostatic water ratio, maintaining a sterile environment, and using gentle handling techniques, you ensure that your research is built on a foundation of integrity and accuracy.
At BioGenix Peptides, we are committed to providing the highest quality materials for your lab. Now that you know the science behind the prep, you’re ready to shop high-quality research peptides and take your experiments to the next level. Happy researching!