The Entero-Mitochondrial Loop: How Gut Peptides Control Cellular Energy Output
The Entero-Mitochondrial Loop: How Gut Peptides Control Cellular Energy Output
Research & Educational Use Only
The information in this article is intended solely for theoretical research discussion and educational purposes. It does not constitute medical, clinical, or therapeutic advice. Any compounds mentioned are discussed in the context of laboratory, in-vitro, and non-human research only and are not approved for diagnosis, treatment, or prevention of any disease.
How gut-derived peptide signaling may act as a hidden master regulator of mitochondrial performance
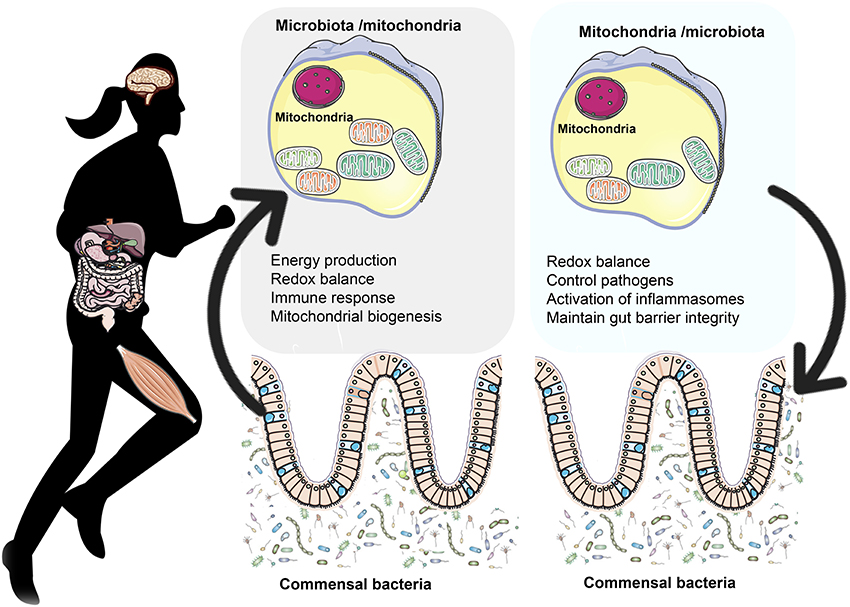
In the peptide research world, mitochondria are usually treated as the endpoint of the story: the place where ATP is produced, oxidative stress is managed, and cellular energy output is determined. But a growing body of theoretical and experimental work points to something deeper:
Gut-derived peptide signals may play a central role in how mitochondria behave in cells across the entire body.
This idea forms the basis of what we’ll call the Entero-Mitochondrial Loop — a bidirectional communication axis in which:
- The gut senses nutrient and environmental status
- It releases signaling peptides into circulation
- Those peptides interact with receptors in tissues
- Mitochondria adjust energy production, substrate use, and stress responses accordingly
In this article, we’ll explore several key peptide players often discussed in research: MOTS-C, Humanin, 5-Amino-1MQ, and GLP-1 analogs (and related multi-agonists). The goal is not to make clinical claims, but to map out a theoretical model of how gut peptide signaling may interact with mitochondrial function.
What Is the “Entero-Mitochondrial Loop”?
The term entero-mitochondrial loop describes a proposed feedback system between:
- Entero- (gut, especially enteroendocrine cells and gut-associated tissues)
- Mitochondria (the cellular powerhouses embedded in virtually every tissue)
In this model, gut-derived peptides and mitochondrial-derived peptides talk to each other through circulation and receptor signaling. The loop can be summarized in four conceptual steps:
- Input: The gut senses nutrients, toxins, mechanical stretch, microbial signals, and stress.
- Signaling: Gut and endocrine cells release peptides and hormones into the bloodstream.
- Response: Tissues receive those signals, altering mitochondrial function, substrate preference, and oxidative balance.
- Feedback: Mitochondria generate their own signaling molecules (including mitochondrial-derived peptides) that inform systemic responses and potentially modulate gut signaling in return.
Next, we’ll look at how specific research peptides fit inside this loop.
MOTS-C: The Mitochondria-Encoded “Gut Messenger”
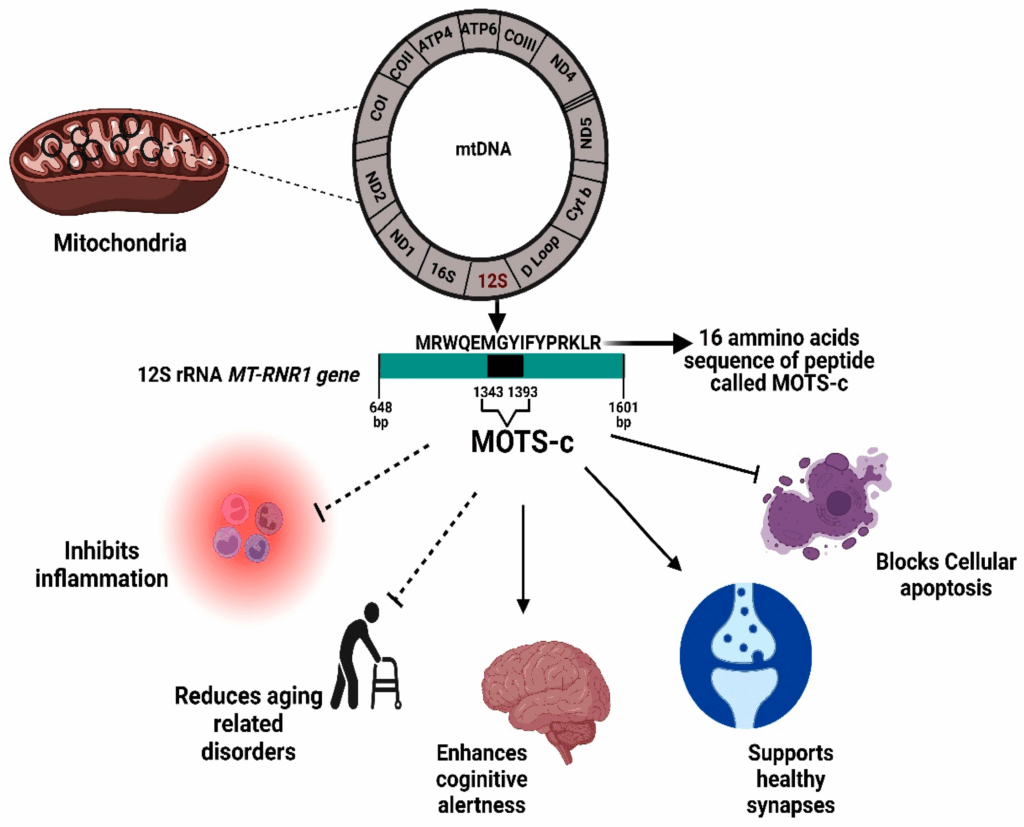
MOTS-C (Mitochondrial ORF of the 12S rRNA type-C) is one of the most intriguing peptide signals in current research because it is encoded in mitochondrial DNA itself. Unlike most proteins and peptides, which come from nuclear DNA, MOTS-C arises from the mitochondria’s own genome.
Experimental data suggest that MOTS-C is responsive to changes in:
- Glucose availability
- Fatty acid levels
- Metabolic stressors such as exercise or nutrient deprivation
One of the key pathways associated with MOTS-C is AMPK (AMP-activated protein kinase), a central energy sensor in cells. When AMPK is activated in animal and cell models, it tends to:
- Increase mitochondrial biogenesis
- Enhance fatty acid oxidation
- Improve glucose uptake
- Support metabolic flexibility during stress
In the context of the entero-mitochondrial loop, researchers have proposed a model where MOTS-C:
- Acts as a systemic signal of mitochondrial and nutrient status
- Interacts with tissues including the gut and skeletal muscle
- Helps coordinate energy use and resilience under metabolic stress
Put simply, MOTS-C can be imagined as a mitochondria-encoded “reporter peptide” that participates in the conversation between nutrient intake (sensed in part by the gut) and cellular energy production (regulated by mitochondria).
Humanin: Cytoprotective Signaling Along the Gut–Brain–Mitochondria Axis
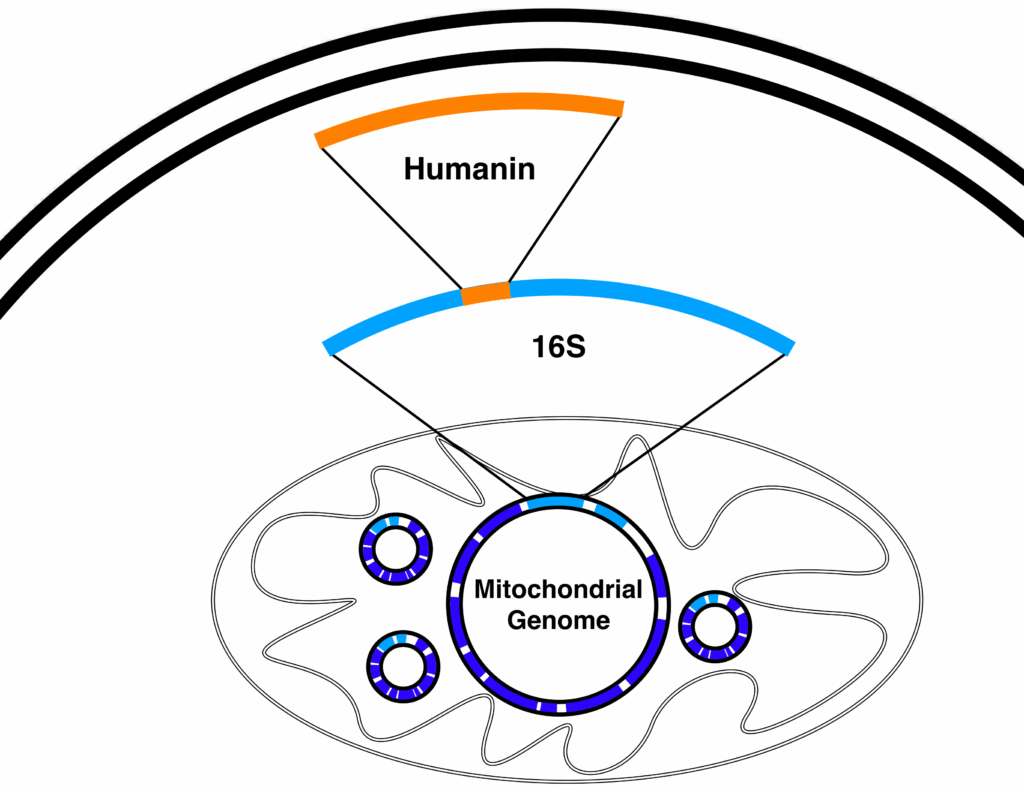
Humanin is another peptide encoded by mitochondrial DNA and has drawn significant interest for its potential cytoprotective effects in preclinical research. Studies suggest that Humanin may help cells handle oxidative and metabolic stress, and it appears in tissues that include the brain, vasculature, and the gastrointestinal tract.
From a theoretical standpoint, Humanin may participate in the entero-mitochondrial loop in several ways:
- Stress buffering: Supporting cells under conditions of oxidative stress, helping preserve mitochondrial integrity.
- Neuro-gut signaling: Potentially influencing the gut–brain axis, where central nervous system signals and gut-derived peptides coordinate metabolic responses.
- Inflammatory tone: Modulating pathways related to inflammatory signaling, which are tightly linked to mitochondrial function and gut barrier integrity.
In this model, Humanin can be imagined as an “SOS peptide” released by mitochondria under stress. That signal may feed into gut and brain circuits that, in turn, adjust systemic energy allocation and mitochondrial behavior across different tissues.
5-Amino-1MQ: NAD⁺ and the Metabolic Throughput Gate
5-Amino-1MQ is not a naturally occurring mitochondrial-derived peptide, but it often appears in research discussions due to its relationship with NAD⁺ metabolism. NAD⁺ is an essential cofactor in redox reactions and mitochondrial respiration.
Although research is still emerging, the interest around 5-Amino-1MQ comes from the idea that modulating NAD⁺ pathways could influence:
- Cellular energy throughput and ATP generation
- Lipid and glucose metabolism
- Balance between anabolic and catabolic processes
- Resilience under metabolic stress
The entero-mitochondrial relevance is indirect but compelling. Gut-derived hormones and peptides can shift the balance of:
- Substrate use (carbohydrates vs. fats)
- Insulin sensitivity
- Systemic inflammatory tone
All of these factors interact with cellular NAD⁺ consumption and regeneration. From a theoretical perspective, 5-Amino-1MQ sits at the “throughput gate,” where mitochondrial energy production and NAD⁺ cycling meet the upstream regulatory signals coming from the gut and endocrine system.
GLP-1, GIP & Multi-Agonists: Gut Hormones That Reprogram Mitochondria
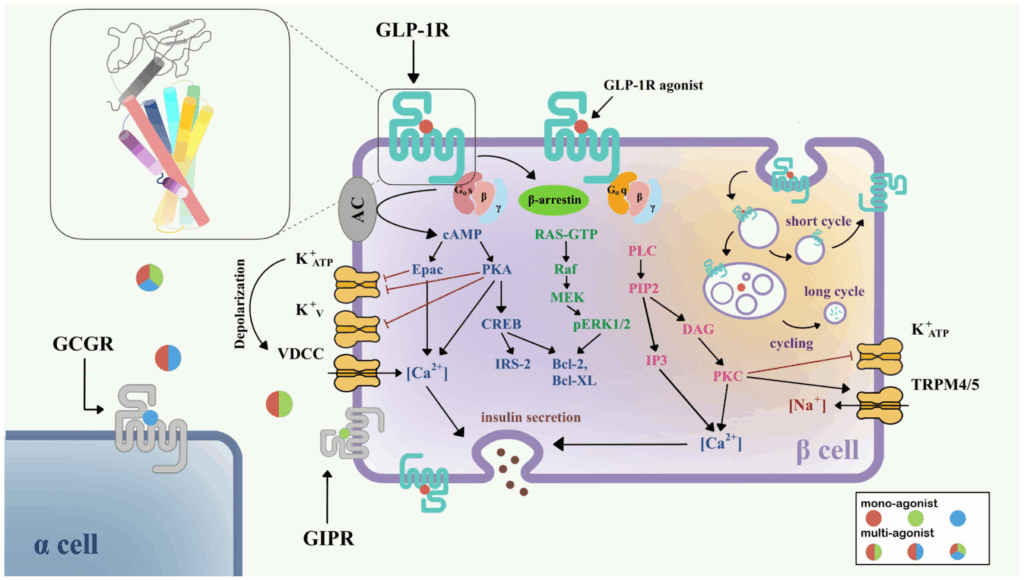
GLP-1 analogs and newer incretin-based multi-agonists have gained attention for their effects on appetite, body weight, and glycemic control in clinical settings. From a research perspective, they also have deep implications for mitochondrial biology.
Gut-derived incretin peptides such as GLP-1 and GIP are released in response to nutrient intake and can influence:
- Insulin secretion and pancreatic function
- Glucagon dynamics
- Gastric emptying and appetite signaling
- Energy expenditure and substrate handling in peripheral tissues
Emerging research suggests that GLP-1 receptor activation may be associated with:
- Improved mitochondrial efficiency in metabolic tissues
- Reduced ectopic lipid accumulation in muscle and liver
- Modulation of oxidative stress and redox balance
- Enhanced insulin sensitivity and metabolic flexibility
Multi-agonist peptides that combine GLP-1 with GIP and/or glucagon signaling further extend this concept, potentially amplifying the entero-mitochondrial message: “Adjust energy production, clear excess fuel, and adapt to the current nutrient environment.”
Putting It All Together: A Research Model of the Entero-Mitochondrial Loop
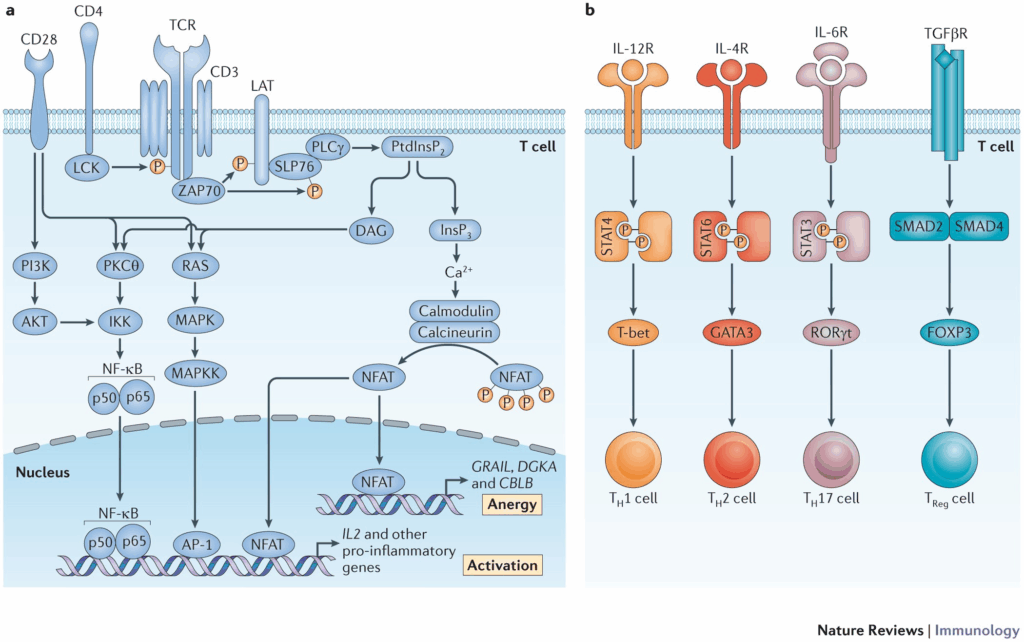
When we place MOTS-C, Humanin, 5-Amino-1MQ, and GLP-1–type peptides into a single framework, a conceptual loop emerges:
Step 1: The Gut Senses the Environment
Nutrient intake, fasting, mechanical stretch, microbiome activity, and stressors such as heat, cold, or training load are detected by the gut. Enteroendocrine cells translate this information into peptide and hormone release.
Step 2: Peptide Messengers Enter Circulation
Peptides such as GLP-1 and GIP (and their analogs) circulate to the pancreas, liver, muscle, adipose tissue, and brain. Meanwhile, mitochondria in various tissues may release peptides like MOTS-C and Humanin in response to internal stress or nutrient status.
Step 3: Mitochondria Receive “Instruction Sets”
Through downstream signaling, mitochondria adjust:
- Biogenesis: How many mitochondria are produced.
- Substrate preference: Whether cells lean more heavily on fats vs. carbohydrates.
- Oxidative stress handling: Antioxidant defenses and redox balance.
- Insulin sensitivity: How cells respond to insulin and manage glucose uptake.
- Energy throughput: ATP output and NAD⁺-related metabolism.
Step 4: Feedback to the Gut and Brain
As mitochondria adapt, they can generate signals back to the rest of the body—ranging from metabolic byproducts and ROS patterns to mitochondrial-derived peptides like MOTS-C and Humanin. These signals may, in turn, influence gut hormone release, inflammatory tone, and even behavior (for example, appetite and fatigue perception).
This theoretical entero-mitochondrial loop is not a single pathway but a dense network of feedback mechanisms where gut peptides and mitochondrial signals co-regulate the body’s overall energy strategy.
Why the Entero-Mitochondrial Loop Matters for Peptide Research
For advanced peptide researchers, the entero-mitochondrial loop concept offers a unifying way to think about:
- Why peptides that “live” in different categories (gut hormones, mitochondrial peptides, NAD⁺-linked compounds) may converge on similar metabolic outcomes.
- How nutrient timing, fasting, and environmental stress (heat, cold, training) can change the way peptides are expressed and how strongly they signal.
- Why the same compound may appear to behave differently depending on baseline metabolic state, body composition, and prior stress history of the research subject.
Instead of viewing each peptide in isolation, the entero-mitochondrial loop encourages a systems-level perspective: peptides as part of a signaling ecosystem that regulates how mitochondria allocate energy and respond to stress throughout the organism.
Key Research Themes & Search Topics Around the Entero-Mitochondrial Loop
For educational platforms and scientific content hubs, this topic opens up a wide range of high-intent research queries, including:
- “Mitochondrial peptides and metabolism”
- “MOTS-C and metabolic flexibility research”
- “Humanin peptide and cellular stress responses”
- “GLP-1 analogs and mitochondrial function”
- “NAD⁺, 5-Amino-1MQ, and energy metabolism”
- “Gut–mitochondria axis in metabolic research”
- “Entero-mitochondrial communication loop”
Content built around these themes can help position a brand or educational resource as science-driven, emphasizing mechanistic pathways and preclinical data rather than clinical or therapeutic claims. Comprehensive Research & Compliance Disclaimer
The concepts discussed in this article, including the “entero-mitochondrial loop” and the roles of peptides such as MOTS-C, Humanin, 5-Amino-1MQ, and GLP-1–related analogs, are presented for theoretical and educational discussion only. Any mechanisms, pathways, or effects described relate to preclinical or experimental research contexts and must not be interpreted as established clinical outcomes.

MOTS-c 10mg
MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c) is a 16–amino-acid peptide encoded within the mitochondrial 12S rRNA gene region. It belongs to the family of mitochondrial-derived peptides (MDPs) that function as retrograde signals, coordinating communication between mitochondria and the nucleus to influence metabolic and stress-response pathways. Under metabolic stress—such as glucose restriction or elevated oxidative load—MOTS-c has been observed to translocate to the nucleus, where it can modulate gene expression and support adaptive metabolic regulation.
In Stock
Humanin 10mg
Humanin is a small 24–amino-acid peptide that comes from the mitochondria, the parts of the cell responsible for energy production. Researchers study Humanin because it appears to help protect cells during stress, support healthy energy balance, and promote normal metabolic function.
Early studies suggest Humanin may help shield cells from oxidative damage, support healthier mitochondrial activity, and influence pathways related to aging, energy use, and cell survival. It has also been explored for potential roles in brain health, insulin sensitivity, and general cellular resilience.
Humanin is part of a group of naturally produced “mitochondrial peptides” that may act as internal signals, helping the body respond to stress and maintain normal cellular function under challenging conditions.
Out of stock
Reta (GLP-3) 30mg
Retatrutide is an investigational multi-agonist peptide engineered to activate three metabolic hormone receptors—GLP-1, GIP, and glucagon—positioning it as a next-generation “triple agonist.” In research settings, it has been examined for its potential influence on appetite regulation, energy utilization, and body-weight–related endpoints. Chemically, retatrutide is a long-acting incretin-mimetic designed to co-activate GLP-1R, GIPR, and GCGR, enabling coordinated engagement of complementary metabolic pathways. Studies have explored its combined effects on glucose homeostasis, gastric emptying, insulin dynamics, lipolysis, and energy expenditure within controlled experimental models.
In Stock
NAD+ 500mg
NAD⁺ (Nicotinamide Adenine Dinucleotide) is a molecule found in every cell of the body and is essential for creating energy. Researchers study NAD⁺ because it helps power the mitochondria—the parts of the cell responsible for energy production—and supports processes related to metabolism, cellular repair, and overall longevity.
In research settings, NAD⁺ has been explored for its role in maintaining healthy energy levels, supporting DNA repair, and helping cells function more efficiently as they age. Many studies also focus on how NAD⁺ levels naturally decline over time, making it an important molecule for understanding aging, vitality, and long-term cellular health.
In Stock




One thought on “The Entero-Mitochondrial Loop: How Gut Peptides Control Cellular Energy Output”
Justin Ziemniak
January 21, 2026JZ JZ JZS