Tesamorelin: Beyond Growth Hormone — What the Research Really Shows
Research Disclaimer (Educational Use Only):
This article is provided for educational and informational purposes only and summarizes published scientific literature. It does not constitute medical advice, diagnosis, or treatment guidance. Any compounds referenced are intended strictly for laboratory, analytical, and research use only and are not for human or animal consumption.
Introduction
Tesamorelin is often described as a “growth hormone peptide,” but the research story is more nuanced—and more interesting. Rather than acting like exogenous growth hormone, tesamorelin is a GHRH (growth hormone–releasing hormone) analogue that stimulates pulsatile endogenous GH release through the pituitary, with downstream effects that have been studied most rigorously in visceral adiposity and metabolic risk biology. In this article
- What Tesamorelin is (and what it isn’t)
- How it works: GHRH receptor → GH pulses → IGF-1
- Why visceral fat is the “signature” outcome
- Liver fat & metabolic signaling: the adipose–liver axis
- Body composition and tissue quality (what imaging reveals)
- Metabolic markers and cardiometabolic context
- Endocrine rhythm & physiology-first signaling
- Tolerability and research monitoring (high-level)
- FAQ
- Peer-reviewed references
1) What Tesamorelin Is (and What It Isn’t)
Tesamorelin is a synthetic analogue of GHRH designed to activate the GHRH receptor in the pituitary. In clinical research, this approach is meaningfully different from administering growth hormone directly: it aims to promote a more physiology-aligned pattern of GH secretion—often described as pulsatile release—while still engaging the GH/IGF-1 axis.
Research framing
- Upstream endocrine signaling (GHRH receptor activation)
- Downstream GH pulses → IGF-1 response
- Most-studied outcomes: visceral adiposity (VAT) and related metabolic markers
What it’s not
- Not exogenous GH “replacement”
- Not a guaranteed “scale-weight” agent (VAT changes can occur without dramatic net weight change)
- Not a substitute for clinical oversight in medical contexts
2) How It Works: GHRH Receptor → GH Pulses → IGF-1
Mechanistically, tesamorelin binds the GHRH receptor on pituitary somatotroph cells, stimulating secretion of growth hormone. GH then signals in peripheral tissues—most notably the liver—to increase IGF-1, a key mediator of GH’s systemic effects. In research contexts, this endocrine cascade is often discussed as a pathway that can influence: lipolysis, fat distribution, liver lipid handling, and tissue remodeling signals. Simplified pathway map (education-first) Tesamorelin GHRH analogue Pituitary (GHRH-R) ↑ Pulsatile GH release Peripheral response ↑ IGF-1 + metabolic signaling
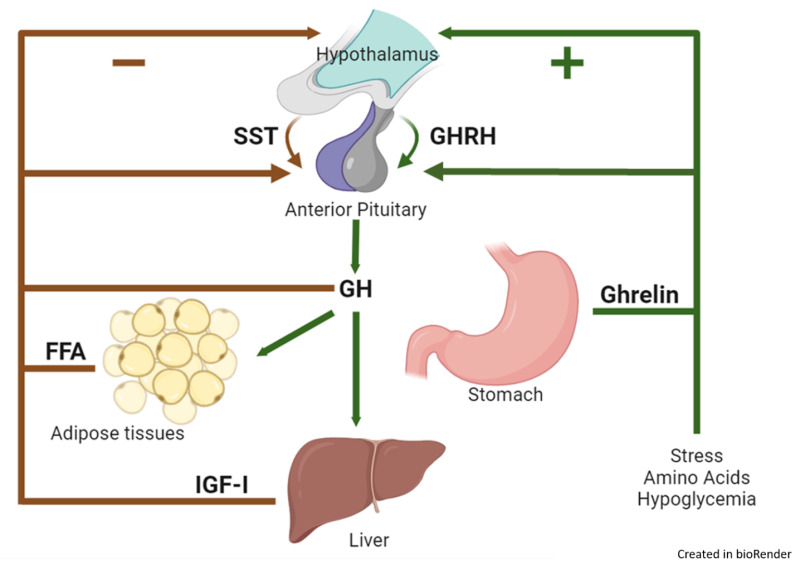
3) Why Visceral Fat Is the “Signature” Outcome
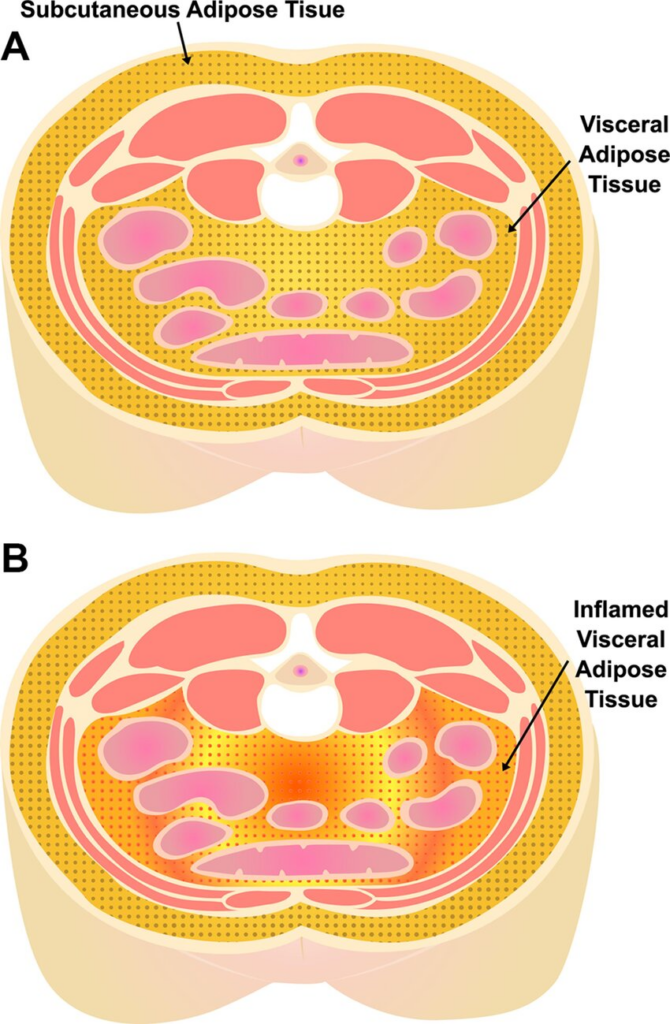
The strongest clinical evidence base for tesamorelin comes from studies in HIV-associated lipodystrophy, where researchers used imaging-based endpoints (CT/MRI) to quantify changes in visceral adipose tissue (VAT). Across randomized controlled trials, tesamorelin was associated with clinically meaningful reductions in VAT compared with placebo, even when overall scale weight did not shift dramatically.
Why VAT matters (systems view)
- VAT is metabolically active and more strongly associated with insulin resistance and cardiometabolic risk than subcutaneous fat.
- VAT communicates with the liver through portal circulation, influencing hepatic lipid handling and inflammatory tone.
- Reducing VAT can correlate with improvements in selected metabolic markers, depending on population and study design.
Importantly, these studies used objective imaging rather than relying only on body weight. That’s a big deal: weight can be “noisy,” and visceral compartment changes may not always track perfectly with the scale.
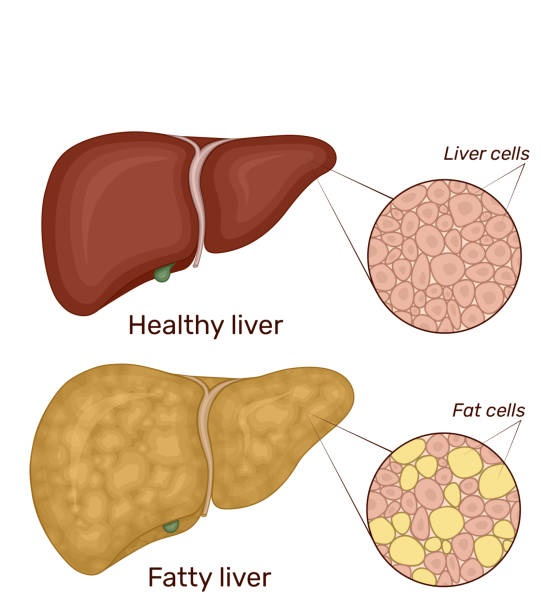
4) Liver Fat & Metabolic Signaling: The Adipose–Liver Axis
Beyond VAT, research has explored tesamorelin’s relationship with hepatic fat content—especially in people with HIV who also have nonalcoholic fatty liver disease (NAFLD). In controlled trials, tesamorelin has been associated with reductions in liver fat, supporting the idea that changing visceral adiposity and endocrine signaling can influence downstream liver biology.
How researchers interpret this
A useful way to think about this is “cross-talk”: visceral adipose tissue, growth hormone dynamics, and liver lipid metabolism are interlinked. When VAT decreases and hormonal signaling shifts, liver fat handling may shift too—especially in susceptible metabolic contexts.
5) Body Composition & Tissue Quality: What Imaging Reveals
Another reason tesamorelin remains a standout in peptide research is the quality of measurement used in many studies: CT-based analysis can quantify not only “how much” tissue exists, but also where it is and (in some protocols) infer aspects of tissue density/quality.
What researchers often measure
- Visceral adipose tissue area/volume
- Subcutaneous adipose tissue compartments
- Trunk fat distribution changes
- Selected muscle cross-sectional indices (study-dependent)
Why this is meaningful
- Distribution can matter as much as totals
- Imaging reduces reliance on scale weight alone
- Allows mechanistic hypotheses (adipose compartment biology)
6) Metabolic Markers & Cardiometabolic Context
In the clinical literature, changes in VAT are sometimes accompanied by shifts in metabolic markers (for example, triglycerides or certain inflammatory or risk-related measures). These effects can vary by population, baseline risk, diet/activity controls, and endpoints. A careful reading of the evidence suggests tesamorelin is best understood as a compound with: fat-distribution effects plus downstream metabolic signaling implications, rather than a one-dimensional “weight loss” narrative.
| Domain | What the research often emphasizes | Why it matters |
|---|---|---|
| Visceral fat (VAT) | Imaging-based VAT reduction in specific clinical populations | VAT is strongly linked to cardiometabolic risk biology |
| Liver fat | Changes in hepatic fat content in targeted trials | Liver fat is central to metabolic syndrome progression |
| IGF-1 | Physiologic axis engagement (monitoring relevance) | IGF-1 sits at the intersection of growth and metabolic signaling |
7) Endocrine Rhythm: A Physiology-First Signaling Style
A recurring theme in endocrinology research is that timing and pulsatility matter. Growth hormone is naturally secreted in pulses, influenced by sleep and neuroendocrine inputs. Because tesamorelin operates through the GHRH receptor, researchers often discuss it as a “rhythm-respecting” approach—one that stimulates endogenous secretion rather than bypassing the axis.
Plain-language takeaway
Think of tesamorelin as a “signal to release” rather than a “replacement hormone.” In research, that distinction is linked to how the body coordinates downstream responses across tissues.
8) Tolerability & Research Monitoring (High-Level)
In the clinical literature, tesamorelin has generally been described as well tolerated, with commonly reported events including injection-site reactions and fluid-related symptoms in some contexts. Because tesamorelin can raise IGF-1, research discussions often include the importance of monitoring IGF-1 and considering population-specific contraindications in medical use contexts. For educational clarity: this article does not provide medical guidance—only a high-level summary of what the published literature discusses.
Research best practice (conceptual)
- Define endpoints clearly (VAT imaging vs scale weight)
- Track axis biomarkers when relevant (e.g., IGF-1 in clinical protocols)
- Interpret outcomes in the context of diet/activity controls and baseline risk
Explore related educational topics
- Research Library
- HGH Secretagogues: A Research Overview
- Receptor Signaling & Downregulation (Why “dose” isn’t the whole story)
FAQ
Is Tesamorelin the same thing as growth hormone?
Not in mechanism. Growth hormone administration supplies GH directly, while tesamorelin is a GHRH analogue that stimulates the pituitary through the GHRH receptor to increase endogenous GH secretion. In research discussions, this difference matters because it involves upstream regulation, feedback loops, and physiologic secretion patterns. Why do studies focus on visceral fat instead of just “weight loss”?
Many tesamorelin trials used CT/MRI imaging to quantify visceral adipose tissue (VAT), a metabolically active fat depot strongly associated with cardiometabolic risk. VAT can change without dramatic shifts in total body weight—so imaging endpoints can reveal effects the scale may miss. Does tesamorelin affect IGF-1?
In clinical research, tesamorelin commonly increases IGF-1 as a downstream effect of stimulating GH release. In medical contexts, monitoring IGF-1 is often discussed as part of oversight. This article is educational and does not provide medical advice. What’s the best way to interpret tesamorelin research as a reader?
Look for (1) imaging-based VAT endpoints, (2) study duration, (3) population and baseline risk, and (4) how diet/exercise were controlled. Those factors strongly influence effect sizes and how transferable findings may be across different research contexts.
Conclusion
Tesamorelin stands out in peptide research because it is supported by a comparatively robust clinical literature focused on visceral fat reduction and related metabolic outcomes, measured using rigorous imaging endpoints. Its upstream endocrine mechanism—GHRH receptor activation leading to pulsatile GH secretion and IGF-1 response—makes it a compelling model for studying how hormonal rhythm and fat distribution influence broader metabolic biology.
For researchers and science-focused readers, the key is to interpret tesamorelin through the lens of compartment biology and systems signaling—not simplistic “weight loss” narratives.
Peer-Reviewed References
- Clinical Review Report: Tesamorelin (Egrifta) — regulatory and literature summary.
- Tesamorelin Improves Fat Quality Independent of Changes in Fat Quantity — CT-based adipose tissue quality study.
- Effect of Tesamorelin on Visceral Fat and Liver Fat in HIV-Infected Patients — GHRH analogue effects on hepatic and visceral fat.
- Visceral Fat Reduction with Tesamorelin Associated with Improved Liver Enzymes — secondary analysis from Phase III trials.
- Falutz et al. — Effects of Tesamorelin in HIV Patients — VAT reduction and body composition outcomes.
- Effects of Tesamorelin (TH9507) — GH-Releasing Factor Analog — long-term VAT maintenance and body composition.
- ClinicalTrials.gov — Registered Tesamorelin Study — ongoing clinical research registry.
- Visceral Fat Reduction Sustained Over 52 Weeks — longitudinal clinical outcome summary.
Final Thought
Tesamorelin is not simply “another GH peptide.”
Its value lies in how selectively and context-dependently it influences growth hormone signaling — and how that signaling ripples into fat metabolism, liver function, and neuroendocrine timing.
As research continues to move away from blunt hormonal manipulation and toward precision signaling, tesamorelin remains a compelling compound to study.

Tesamorelin 10mg
Tesamorelin is a synthetic 44–amino-acid polypeptide modeled on growth hormone–releasing hormone (GHRH), incorporating N-terminal and C-terminal modifications to enhance stability and resistance to enzymatic degradation. It binds to GHRH receptors in the anterior pituitary, promoting growth hormone (GH) secretion and subsequent stimulation of insulin-like growth factor-1 (IGF-1).
Through this pathway, Tesamorelin has been investigated for its potential effects on fat metabolism, lipodystrophy, cognitive parameters, and muscle composition in controlled research settings.
In Stock
Tesamorelin 2mg
Tesamorelin is a synthetic 44–amino-acid polypeptide modeled on growth hormone–releasing hormone (GHRH), incorporating N-terminal and C-terminal modifications to enhance stability and resistance to enzymatic degradation. It binds to GHRH receptors in the anterior pituitary, promoting growth hormone (GH) secretion and subsequent stimulation of insulin-like growth factor-1 (IGF-1).
Through this pathway, Tesamorelin has been investigated for its potential effects on fat metabolism, lipodystrophy, cognitive parameters, and muscle composition in controlled research settings.





