SS-31 (Elamipretide): A Mitochondrial Inner-Membrane Stabilizer
Structural integrity, energetic fidelity, and the foundation of cellular performance (research overview)
Research Use Only Disclaimer: This article is provided for educational and informational purposes only and reflects theoretical and preclinical research discussions related to mitochondrial biology and peptide science. It does not constitute medical advice. All compounds discussed are intended strictly for research, laboratory, and analytical use only and are not for human or animal consumption.
Introduction: Why Mitochondrial Efficiency Matters More Than Output
In metabolic and performance research, mitochondria are often discussed in terms of quantity—biogenesis, density, and stimulation. However, a growing body of research emphasizes that mitochondrial quality and structural integrity may be equally, if not more, important than sheer mitochondrial number.
SS-31 (also known as Elamipretide) represents a distinct class of research peptides designed not to “stimulate” energy production, but to preserve mitochondrial structure and function at a foundational level: the inner mitochondrial membrane. Rather than pushing mitochondria to work harder, SS-31 is studied for helping mitochondria work correctly under stress.
What Is SS-31?
SS-31 is a short, aromatic–cationic tetrapeptide studied for its affinity for the inner mitochondrial membrane, particularly regions enriched in cardiolipin, a phospholipid that helps organize the electron transport chain (ETC).
High-level characteristics (research framing)
- Small molecular size
- Net positive charge (cationic)
- Selective association with mitochondrial membranes
- Membrane-active behavior rather than receptor-driven signaling
A practical implication of this design is that SS-31 can associate with mitochondria even in contexts where membrane function is compromised, because its localization is discussed as less dependent on “perfect” mitochondrial energetics than many other classes of molecules.
The Inner Mitochondrial Membrane: The True Target
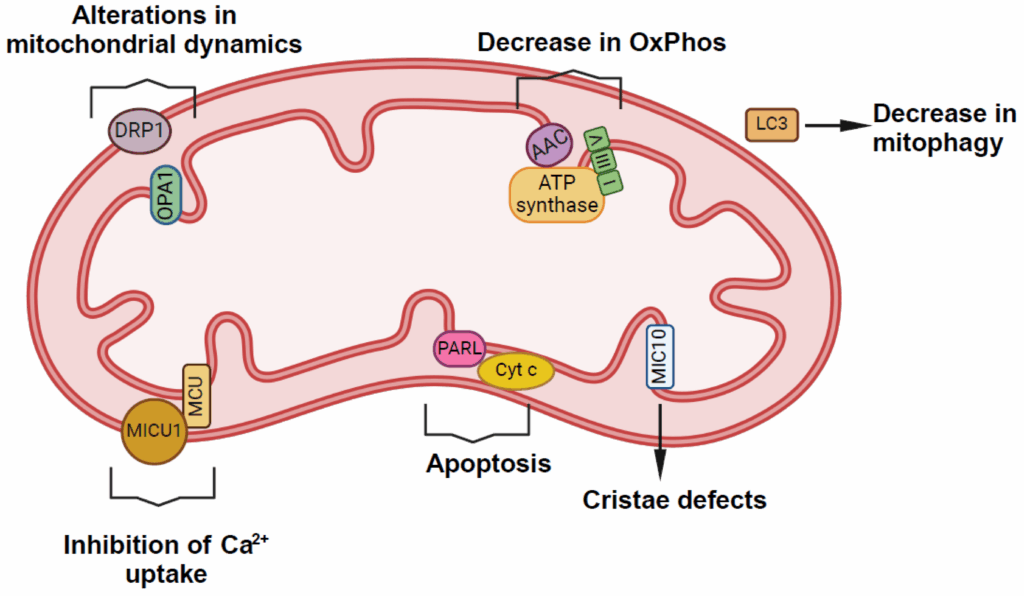
The inner mitochondrial membrane is one of the most functionally dense structures in cell biology. It houses:
- Electron transport chain complexes (I–IV)
- ATP synthase
- Supercomplex organization (functional assemblies of ETC components)
- Proton gradients essential for ATP generation
When this membrane is disrupted, the result is not simply “less energy”—it can distort redox balance, compromise energetic efficiency, and alter how cells interpret stress signals.
Cardiolipin: The Structural Keystone
Cardiolipin is a unique phospholipid found almost exclusively in the inner mitochondrial membrane. It is discussed in research as critical for:
- Anchoring and stabilizing ETC complexes
- Maintaining cristae curvature and membrane architecture
- Supporting efficient electron flow and supercomplex formation
- Reducing destabilization events linked to cytochrome-c release
Under oxidative or metabolic stress, cardiolipin may become peroxidized. This is associated in research models with:
- Electron transport inefficiency
- Increased electron leak
- Higher reactive oxygen species (ROS) generation
- Reduced ATP production efficiency
SS-31 is studied for its ability to bind cardiolipin and help preserve inner-membrane structure, with downstream effects that may include improved ETC organization and reduced lipid peroxidation susceptibility in certain experimental contexts.
Mechanism of Action: Stabilization Over Stimulation
SS-31 is primarily discussed as operating through direct membrane interaction, rather than classic receptor binding. In research framing, the focus is on structural and functional preservation of mitochondrial membranes.
1) Cardiolipin binding and protection
SS-31 is described as associating with cardiolipin-rich regions, potentially supporting membrane resilience and reducing vulnerability to oxidative damage.
2) Electron transport chain efficiency (organization and fidelity)
By helping preserve inner-membrane architecture, SS-31 may support:
- More consistent ETC complex organization
- Reduced electron leak
- Improved proton gradient stability
- More efficient ATP production under stress (without “forcing” demand)
3) Reduction of ROS at the source
SS-31 is often contrasted with traditional antioxidants. Rather than scavenging ROS after formation, it is discussed as potentially reducing ROS generation by improving electron transport fidelity upstream.
Key concept: In research language, SS-31 is better framed as a mitochondrial efficiency stabilizer than a stimulant. It is not typically positioned as a compound that “pushes” output—rather one that may reduce failure modes under stress.
SS-31 vs Traditional “Mitochondrial Boosters”
Many popular mitochondrial agents aim to stimulate signaling pathways (e.g., biogenesis-related transcription) or increase energetic demand. SS-31 stands out because it focuses on the structural platform the ETC depends on.
| Feature | Traditional agents (general) | SS-31 (research framing) |
|---|---|---|
| Primary focus | Upstream signaling / stimulation | Inner-membrane stabilization |
| Site of action | Nuclear/cytosolic pathways | Mitochondrial inner membrane |
| ROS handling | Often downstream scavenging | Potential upstream reduction of generation |
| Energy demand | May increase demand | Neutral / stabilizing |
In short: SS-31 is not typically framed as making mitochondria “stronger” via stimulation. It’s framed as helping mitochondria operate with greater energetic fidelity when conditions would otherwise degrade performance.
Research Contexts Where SS-31 Often Appears

SS-31 is commonly explored in experimental models that involve mitochondrial vulnerability, including:
- Age-associated mitochondrial decline (general research topic)
- Ischemia–reperfusion stress models (energy system disruption)
- Skeletal muscle fatigue and energetic inefficiency contexts
- Cardiac mitochondrial stress models
- Neuroenergetic stress and high-demand tissue research
- Metabolic inefficiency states where redox balance is disrupted
Across these settings, SS-31 is frequently positioned around a common theme: mitochondrial structure-first preservation.
SS-31 and Cellular Performance (Research Framing)
In performance-oriented research discussions, SS-31 is typically framed as a compound of interest for efficiency, resilience, and recovery capacity rather than hypertrophy signaling.
Common investigation angles
- Maintaining ATP output consistency under repeated stress
- Reducing “energetic noise” from excessive ROS
- Supporting recovery between high-demand intervals
- Preserving mitochondrial signaling accuracy under load
Importantly, SS-31 is not generally discussed as directly activating classic muscle growth pathways (e.g., mTOR/IGF-1) in the way many “anabolic” research conversations are framed. Any downstream performance observations are typically interpreted as secondary to improved mitochondrial integrity.
SS-31 and MOTS-C: A Brief Note on “Priming” (Indirect Relationship)
SS-31 is sometimes discussed in advanced research circles as a permissive or “priming” tool for mitochondrial signaling peptides, including MOTS-C. This does not mean SS-31 activates MOTS-C or increases its expression directly.
Instead, the rationale is mechanistic: mitochondrial-encoded signaling peptides (like MOTS-C) operate within and report on mitochondrial status. If inner-membrane integrity is compromised and ROS generation is excessive, mitochondrial-to-nuclear signaling may become distorted. By stabilizing cardiolipin-rich membranes and reducing redox disruption, SS-31 may help support a cellular environment in which mitochondrial signaling outputs are clearer and more reliable.
This relationship is best framed as contextual and indirect—a question of mitochondrial readiness and signal fidelity rather than direct activation.
Why SS-31 Is Often Misunderstood
SS-31 is frequently mischaracterized as a stimulant or a standard antioxidant. In research framing, it is more accurate to describe SS-31 as:
A mitochondrial inner-membrane stabilizer studied for preserving electron transport efficiency and redox fidelity under stress.
That “hardware-first” approach explains why SS-31 continues to appear in advanced mitochondrial research discussions even when it lacks obvious stimulant-style effects.
Research Summary
SS-31 (Elamipretide) represents a structure-first approach to mitochondrial research, emphasizing:
- Inner mitochondrial membrane integrity
- Cardiolipin preservation
- Electron transport chain organization and efficiency
- Reduction of ROS generation linked to electron leak
- Energetic fidelity under stress
Rather than pushing cells harder, SS-31 is studied for helping cells function with greater accuracy when challenged—protecting the mitochondrial platform that many downstream signals depend on.
Peer Reviewed References — SS-31 / Elamipretide
- Karaa A, Haas R, Goldstein A, Vockley J, Weaver WD, Cohen BH, et al. Efficacy and safety of elamipretide in individuals with primary mitochondrial myopathy: the MMPOWER-3 randomized clinical trial. Neurology. 2023. Link
- Karaa A, Haas R, Goldstein A, Vockley J, Weaver WD, Cohen BH, et al. Efficacy and safety of elamipretide in individuals with primary mitochondrial myopathy: the MMPOWER-3 randomized clinical trial. Neurology. 2023. doi: Link
- Karaa A, Haas R, Goldstein A, Vockley J, Weaver WD, Cohen BH, et al. Randomized dose-escalation trial of elamipretide in adults with primary mitochondrial myopathy. Neurology. 2018;90(14):e1212–e1221. doi: Link
- Campbell MD, Duan J, Samuelson AT, Gaffrey MJ, Merrihew GE, Egertson JD, et al. Improving mitochondrial function with SS-31 reverses age-related redox stress and improves exercise tolerance in aged mice. Free Radic Biol Med. 2018. Link
- Thompson WR, Hornby B, Manuel R, Bradley E, Laux J, Carr J, et al. A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome (TAZPOWER). Genet Med. 2021. Link
- Hornby B, McClelland V, Robinson D, Thompson WR, Laux J, Finsterer J, et al. Natural history comparison study to assess the efficacy of elamipretide in Barth syndrome. Orphanet J Rare Dis. 2022. Link
- Campbell MD, Duan J, Samuelson AT, et al. Intermittent treatment with elamipretide preserves exercise tolerance and mitochondrial outcomes in a preclinical model. 2023. Link
- ClinicalTrials.gov. A Trial to Evaluate Safety and Efficacy of Daily Subcutaneous Elamipretide in Primary Mitochondrial Myopathy (MMPOWER-3). Identifier: NCT03323749. Link
- ClinicalTrials.gov. A Trial to Evaluate Safety, Efficacy, and Tolerability of Elamipretide in Subjects with Barth Syndrome. Identifier: NCT03098797. Link

SS-31 50mg
In Stock
SS-31 10mg
In Stock