Peptides for Dental Regeneration: Emerging Research on Gum Repair, Implant Healing, and Enamel Restoration
Research & Educational Notice:
The information presented in this article is provided strictly for scientific discussion and educational purposes related to peptide research, regenerative biology, and molecular signaling. The material below discusses experimental findings from published scientific literature involving peptides and signaling molecules being studied in laboratory, preclinical, and select dental research settings. This content does not constitute medical advice, dental advice, diagnosis, or treatment guidance. BioGenix Peptides LLC is not a medical or dental provider, and the compounds discussed are not approved medications for dental treatment. All BioGenix products are intended strictly for laboratory, research, and analytical use only and are not for human or veterinary consumption.
The future of oral health research may be shaped not only by better dental materials and surgical techniques, but also by a deeper understanding of the biological signals that guide healing. In dentistry, researchers are increasingly exploring peptides and related signaling molecules that may influence tissue remodeling, inflammation, antimicrobial defense, and bone regeneration within the oral environment.
Traditional dental care often focuses on restoring damaged structures after problems appear. Regenerative research, by contrast, asks a different question: can biological repair processes be better understood and supported at the molecular level? This is where peptides have generated growing scientific interest. Rather than acting merely as structural materials, many peptides function as signaling molecules that influence how cells communicate during tissue repair and regeneration.
In the context of oral health, that question becomes especially important. The mouth is one of the most demanding healing environments in the body. Tissues must respond to continuous microbial exposure, chewing forces, saliva enzymes, temperature variation, and constant mechanical use. Any molecule capable of influencing healing biology in that setting naturally attracts attention from regenerative researchers.
Why Oral Tissue Healing Is So Complex
Oral tissues do not heal under calm, protected conditions. The gums, periodontal tissues, oral mucosa, and bone of the jaw are exposed to constant physiological stress. Even a small wound in the mouth may encounter bacterial biofilms, friction from food, and repeated movement throughout the day. These factors make oral healing biologically complex and uniquely vulnerable to delayed repair or inflammatory complications.
Researchers studying oral regeneration often focus on several interconnected biological priorities:
- angiogenesis and blood supply
- collagen production and connective tissue repair
- bone remodeling and osseointegration
- microbial balance and host defense
- cell migration during wound healing
- inflammatory signaling control
- extracellular matrix remodeling
Because peptides often operate as signaling messengers within these systems, they are especially compelling research targets. In regenerative dentistry, the interest is not simply whether a molecule exists in tissue, but whether it influences how repair unfolds over time.
I’m Jay Daniel, Founder and CEO of BioGenix Peptides, and through years of hands-on experience in peptide sourcing, purity verification, and laboratory quality control, I’ve seen firsthand how rapidly peptide science is expanding into new areas of research — including fields like regenerative biology and oral tissue repair. As interest grows around topics such as gum regeneration, dental implant healing, and antimicrobial defense peptides, it becomes increasingly important to understand the biological pathways researchers are exploring. In this guide, I’ll walk you through the emerging science behind peptides being studied in dental and oral tissue research, and why these signaling molecules are drawing attention in discussions of regenerative dentistry.
The Biology of Oral Tissue Regeneration
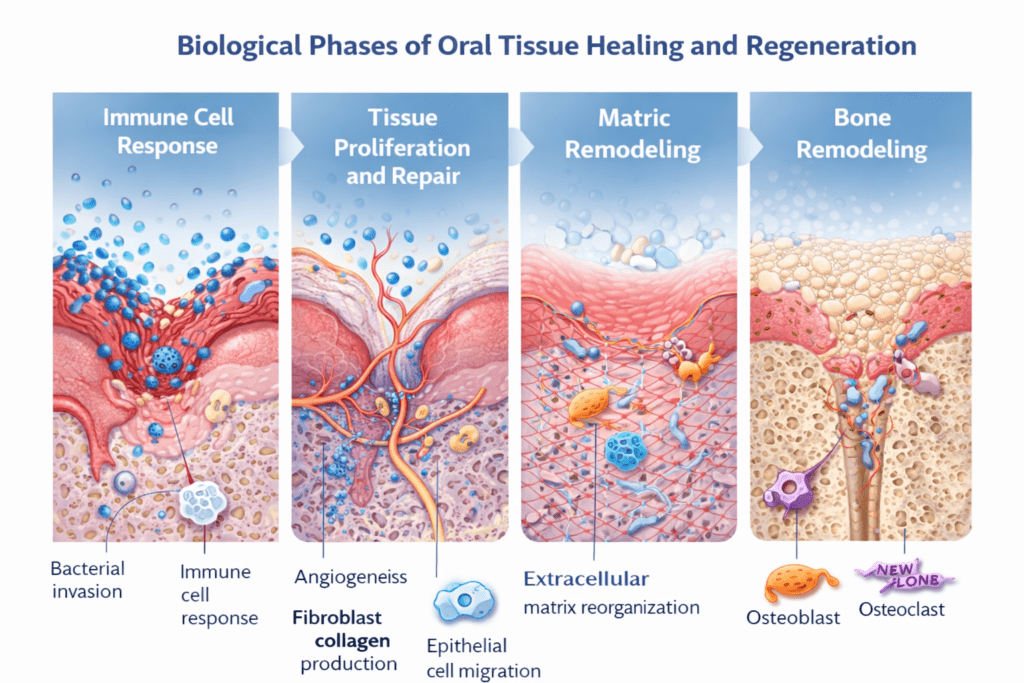
Healing within the oral cavity involves a coordinated sequence of biological phases rather than a single isolated event. After tissue injury, immune cells first respond to microbial exposure and damaged tissue signals. This inflammatory phase is necessary, but it must be controlled. Too little response may impair defense, while too much inflammation can interfere with repair and contribute to tissue breakdown.
Following this early response, the body transitions into a proliferative phase in which fibroblasts, endothelial cells, epithelial cells, and other specialized cell types begin rebuilding tissue architecture. New blood vessels form, connective tissue matrices begin to organize, and surface tissues work to restore protective barriers.
Finally, remodeling processes refine the repaired structure over time. Collagen fibers reorganize, extracellular matrix composition changes, and bone may adapt in response to mechanical loading. This remodeling phase is especially important in dentistry because long-term stability often depends on tissue quality rather than simple wound closure.
Researchers investigating peptides for dental regeneration are often interested in how these molecules interact with one or more of these overlapping phases. A peptide that influences angiogenesis, fibroblast activity, collagen organization, or antimicrobial defense could be relevant not because it “fixes” tissue directly, but because it affects the biological conversation taking place during repair.
How Peptides Work Upstream in Oral Tissue Repair
One of the most interesting aspects of peptide research is that peptides are frequently studied as upstream signaling agents. Rather than only masking symptoms or replacing damaged material, many peptides are investigated for their ability to influence earlier biological events that shape the repair environment itself.
In oral tissue healing, this upstream perspective matters because successful outcomes depend on the quality of early cellular communication. If inflammatory signaling becomes excessive, tissues may break down. If blood supply is inadequate, repair slows. If microbial balance is disrupted, healing becomes more difficult. If connective tissue organization is poor, long-term structural stability may suffer.
Researchers therefore study peptides in relation to pathways involving:
- nitric oxide signaling
- angiogenic communication
- fibroblast activation
- epithelial migration
- collagen turnover
- immune modulation
- bone-forming cellular differentiation
This upstream framework also fits neatly with the broader concept of biomolecular dentistry, in which tissue repair is understood not merely as a mechanical event, but as a dynamic cellular process directed by molecular signals.
Angiogenesis and Blood Supply in Oral Healing
Adequate blood supply is one of the most important determinants of healing quality in dental tissues. New blood vessel formation, known as angiogenesis, allows oxygen, nutrients, growth factors, and immune cells to reach damaged areas. Without sufficient vascular support, both soft tissue healing and bone remodeling may be compromised.
In dental research, angiogenesis is especially important after procedures such as extractions, grafting, periodontal surgery, and implant placement. Each of these situations requires tissues to rebuild under demanding conditions. Vascular signaling influences how effectively the tissue can support repair and how quickly the microenvironment transitions from injury to regeneration.
Experimental interest in BPC-157 partly stems from its association in preclinical literature with pathways related to vascular signaling, endothelial function, and nitric oxide modulation. These are the types of pathways that researchers consider highly relevant when evaluating tissue recovery environments.
Because gingival and mucosal tissues are richly vascularized and highly active biologically, any research molecule associated with angiogenic signaling naturally becomes relevant to discussions of oral repair. This does not mean clinical conclusions should be assumed, but it does explain why peptide-based research continues to attract interest in dental biology.
Osseointegration and Implant Stability
Dental implants rely on a biological process called osseointegration. This refers to the direct structural and functional connection that forms between living bone and the implant surface. Without successful osseointegration, an implant may fail to achieve long-term stability.
Osseointegration is not a simple “bone grows on metal” event. It depends on a carefully coordinated series of biological responses involving inflammatory cells, osteoblasts, osteoclasts, endothelial cells, fibroblasts, and extracellular matrix proteins. The early healing environment around the implant determines whether bone remodeling proceeds in a stable and organized way.
From a regenerative research perspective, implant healing is therefore a molecular question as much as a mechanical one. Researchers evaluate how blood supply, inflammatory regulation, connective tissue adaptation, and bone signaling all interact to support integration.
This is one reason molecules associated with tissue repair and cellular migration remain of interest. TB-500, a synthetic form related to Thymosin Beta-4 signaling, has been discussed in regenerative science because of its relationship to cellular migration, actin regulation, and tissue organization during healing. In oral tissue research, these kinds of processes are directly relevant to how cells repopulate and stabilize a surgically altered environment.
Implant stability is ultimately dependent on more than hardware design alone. It is shaped by the tissue response surrounding the implant, which is why regenerative dentistry increasingly investigates molecular factors that may influence healing quality from the beginning.
The Oral Microbiome and Host Defense Peptides
The mouth contains one of the most diverse and active microbial ecosystems in the human body. Dental plaque, saliva, mucosal surfaces, periodontal pockets, and tongue tissues all host dynamic microbial communities. These communities are not inherently harmful, but their balance plays a major role in oral health and disease.
For healing to proceed efficiently, tissues must maintain a functional relationship with this microbial environment. That is particularly important after extraction, surgery, periodontal therapy, or implant placement, when exposed tissues may be more vulnerable to inflammatory disruption.
Host defense peptides are therefore a major focus in oral biology research. Among the most widely studied is LL-37, an antimicrobial peptide involved in innate immune defense. LL-37 has been studied in relation to microbial membrane interactions, inflammatory signaling, mucosal defense, and oral host-microbe balance.
Its relevance in regenerative dentistry lies in the fact that healing is never just about tissue growth. It is also about the tissue’s ability to defend itself and maintain an environment conducive to repair. If microbial pressure becomes excessive or immune signaling becomes dysregulated, regeneration may be impaired.
This is why antimicrobial and host-defense peptides occupy an important place in modern oral research. They highlight the reality that oral regeneration depends not only on rebuilding tissue, but also on managing the ecological and immune context in which that rebuilding occurs.
Collagen Signaling and Connective Tissue Repair
Healthy oral tissues depend heavily on connective tissue quality. Gums, periodontal ligaments, oral mucosa, and supporting structures all rely on collagen-rich extracellular matrices to provide strength, flexibility, and structural organization. Repair of these tissues requires not only cell proliferation but coordinated matrix rebuilding.
Fibroblasts play a central role in this process by producing collagen and organizing connective tissue architecture. Because of this, research molecules associated with collagen signaling and extracellular matrix remodeling attract attention in discussions of periodontal support and gum regeneration.
GHK-Cu is a naturally occurring copper-binding peptide found in plasma and saliva, and it has been extensively studied in regenerative biology literature for its relationship to tissue remodeling, gene signaling, fibroblast function, and collagen-associated repair pathways.
Its relevance in oral tissue research is straightforward: gum tissues and peri-implant soft tissues must maintain both flexibility and integrity under continuous stress. A signaling molecule associated with connective tissue organization may therefore be of significant interest in preclinical discussions of oral regeneration.
Collagen remodeling is also a long-term issue, not just an early wound healing event. Even after superficial closure occurs, tissue quality continues evolving. This makes collagen signaling a key topic in any deeper exploration of peptides for dental health and implant healing.
BPC-157 and Oral Tissue Repair Research
BPC-157 has been studied in a variety of experimental models related to wound repair, inflammatory signaling, vascular function, and tissue protection. Although much of the broader literature is not specific to dentistry, some preclinical work has explored its relevance to periodontal inflammation and oral injury contexts.
Within research discussions, BPC-157 is often associated with pathways involving:
- angiogenesis-related signaling
- nitric oxide modulation
- cellular migration
- inflammatory response regulation
- tissue-protective repair environments
These are exactly the kinds of pathways that matter in gum healing, mucosal recovery, and post-procedural oral repair models. In that sense, BPC-157 fits naturally into broader discussions of regenerative dentistry, especially where vascular support and early tissue coordination are central concerns.
Its appeal in regenerative research is not simply that it is linked to “healing,” but that it appears in literature tied to the biology that underlies healing. For a topic like oral regeneration, that distinction matters. Serious molecular research focuses on mechanism, not just outcome labels.

BPC-157 10mg
BPC-157, or Body Protection Compound-157, is a synthetic pentadecapeptide derived from a protective protein found in gastric juice. Originally isolated from gastric secretions, this 15-amino acid sequence has been extensively studied in preclinical research for its potential effects on tissue repair, angiogenesis, and cellular protection mechanisms. Research interest centers on its interaction with growth factor pathways and its reported influence on wound healing processes at the molecular level.
GHK-Cu and Gum Regeneration
GHK-Cu occupies an especially interesting place in discussions of oral repair because it exists naturally within human biology and has been studied in relation to multiple repair-associated systems. Research has linked it to gene expression patterns related to tissue remodeling, anti-inflammatory balance, collagen dynamics, and regenerative signaling.
In the setting of oral tissue health, this makes GHK-Cu particularly relevant for conversations about:
- gum tissue integrity
- connective tissue remodeling
- fibroblast-associated repair activity
- collagen-supportive signaling
- soft tissue environments around implants
Because saliva and oral tissues are already part of the peptide’s natural biological context, GHK-Cu carries a certain intuitive relevance in regenerative dentistry discussions. It represents the broader scientific idea that oral healing may be influenced by endogenous signaling molecules involved in maintenance and repair.
GHK-Cu 100mg
GHK-Cu is a naturally occurring copper-binding tripeptide composed of glycyl-L-histidyl-L-lysine chelated to copper (II). It is present in plasma and is released at injury sites, where it is proposed to influence extracellular matrix synthesis and tissue repair. Reported circulating concentrations decline with age (e.g., ~200 ng/mL at ~20 years to ~80 ng/mL by ~60 years). In vitro and in vivo studies have explored GHK (with and without copper) for roles spanning collagen dynamics, antioxidant defenses, wound healing, gene expression modulation,…
LL-37 and Oral Antimicrobial Defense
LL-37 is one of the most important host-defense peptides studied in oral health research. Produced by epithelial and immune cells, it contributes to innate defense by interacting with microbes, modulating inflammatory signaling, and participating in barrier protection.
In oral microbiology and periodontal research, LL-37 has been discussed in relation to:
- bacterial biofilm interactions
- host immune signaling
- mucosal defense
- periodontal disease environments
- microbial balance during healing
This makes LL-37 especially relevant to the concept of regenerative dentistry because tissue repair in the mouth cannot be separated from microbial control. Oral healing is not merely about rebuilding tissue structure; it is about rebuilding that structure within an active ecological environment.
As a result, peptides involved in innate defense may become increasingly important in how researchers think about oral regeneration and implant success.

LL-37 5mg
LL-37 is a naturally occurring peptide that plays an important role in the body’s first line of defense. It is part of the innate immune system and is studied for its ability to help protect the body from bacteria, support healthy inflammation balance, and assist in tissue repair.
TB-500 and Implant Healing Research
TB-500 is a synthetic form related to the naturally occurring peptide Thymosin Beta-4, which has been widely studied for roles in cellular migration, wound repair, tissue remodeling, and regenerative signaling. One of its most notable mechanistic associations is with actin regulation, a process central to cell movement and structural organization.
In oral and implant-related healing discussions, this is highly relevant. Tissue recovery after surgery depends on coordinated migration of cells into damaged or surgically altered regions. Epithelial closure, connective tissue adaptation, vascular support, and matrix remodeling all depend on cells reaching the right places at the right times.
For this reason, TB-500 is often included in broader regenerative conversations involving:
- cellular migration during healing
- soft tissue repair near implant sites
- recovery following oral surgical trauma
- tissue remodeling environments
The significance of TB-500 in regenerative research lies less in simplistic labels and more in the fact that it points toward a key truth: effective healing is organized, not accidental. Molecular signals help coordinate that organization, and cell movement is one of the most fundamental parts of the process.
TB-500 10mg
TB-500 is a synthetic version of Thymosin Beta-4, a naturally occurring protein found in many tissues throughout the body. Researchers study TB-500 for its potential role in supporting healthy tissue repair, circulation, and recovery following physical stress or injury.
P11-4 and Enamel Regeneration Research
P11-4 is one of the most directly relevant biomimetic peptides discussed in dental research because it was specifically developed for enamel-related applications. Unlike broader healing peptides that may be examined across multiple tissue types, P11-4 is notable for its intended relationship to early enamel remineralization and scaffold-like self-assembly behavior.
Researchers have explored P11-4 in relation to:
- early enamel remineralization
- hydroxyapatite formation support
- biomimetic tooth repair concepts
- non-restorative management of early lesions
Although BioGenix does not carry this compound, it is important to discuss in a serious article on dental regeneration because it demonstrates how peptide research is not limited to wound healing alone. It also extends into biomimetic materials science and the idea that mineralized tissues may one day be supported through more sophisticated molecular scaffolding strategies.
Bone Morphogenetic Proteins and Jawbone Regeneration
Bone Morphogenetic Proteins (BMPs) are not peptides in the narrow product-category sense, but they are central to regenerative dentistry and deserve inclusion in any deeper discussion of implant healing and oral tissue engineering. BMPs are powerful signaling molecules involved in bone formation, stem cell differentiation, and osteogenic pathways.
In oral and maxillofacial research, BMP-related strategies have been studied in contexts such as:
- jawbone regeneration
- socket preservation
- sinus augmentation
- bone graft enhancement
- implant site preparation
- periodontal bone defect repair
The reason BMPs matter in this article is conceptual. They reinforce the broader scientific principle that successful dental repair often depends on signaling molecules guiding cells toward regenerative behavior. Whether the target is soft tissue, enamel, or bone, regenerative dentistry increasingly revolves around biological instruction rather than mere replacement.
The Role of Peptides in Dental Implant Healing Research
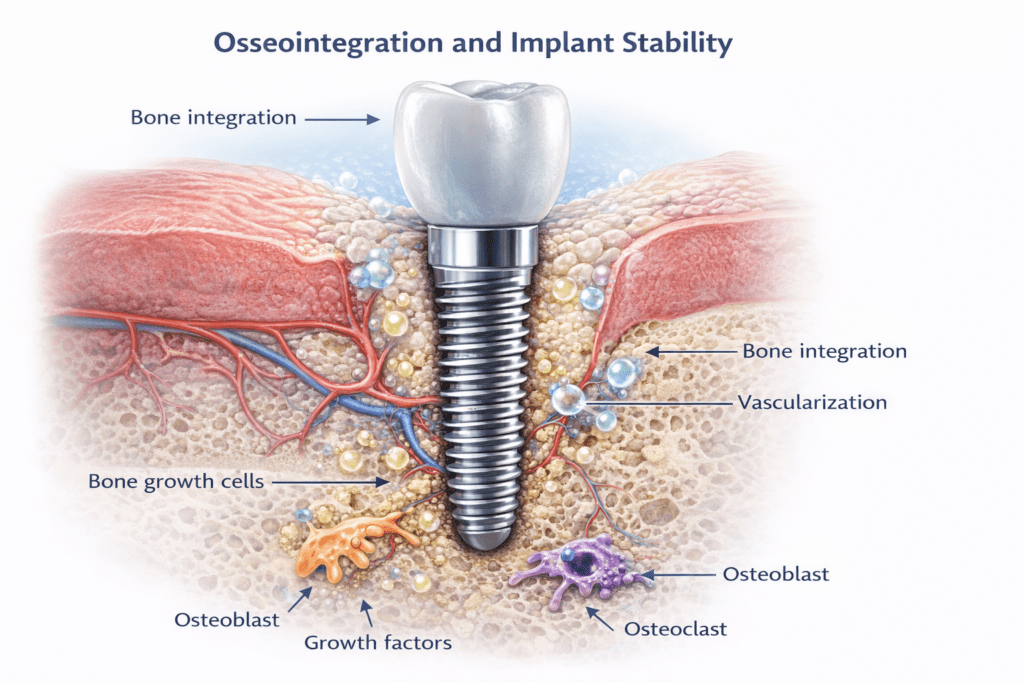
When people search for peptides for dental implant healing, they are often asking a much broader biological question: what molecular signals help tissues stabilize, defend themselves, and integrate around an implanted structure?
The answer is complex because implant healing depends on multiple overlapping systems working together. Among the most important are:
- early inflammatory response coordination
- angiogenesis and blood supply
- fibroblast and connective tissue activity
- bone remodeling and osseointegration
- microbial regulation in peri-implant tissues
- epithelial and soft tissue sealing
This complexity is exactly why regenerative dentistry continues moving toward deeper molecular analysis. The implant itself may be mechanical, but the biological environment around it determines long-term success. That is why peptides and signaling proteins remain such a compelling area of scientific inquiry.
Peptides, Periodontal Biology, and Gum Support
Beyond implants, regenerative researchers are also interested in periodontal tissues. The periodontium includes the gingiva, periodontal ligament, cementum, and supporting alveolar bone. These tissues work together to anchor and protect teeth, yet they are vulnerable to chronic inflammation, microbial imbalance, and structural breakdown.
Gum support and periodontal repair are therefore major themes in peptide-related oral biology. Researchers may examine vascular support, collagen organization, host-defense peptides, inflammatory signaling, and tissue remodeling pathways as part of larger attempts to understand how oral structures maintain resilience or recover from injury.
From this perspective, different peptides may be relevant for different reasons. Some may be discussed for vascular and repair-related pathways, others for connective tissue remodeling, and others for microbial defense. Together, they form a broader scientific picture of oral healing as a networked process rather than a single isolated event.
The Emerging Field of Biomolecular Dentistry
The growing interest in peptides for dental regeneration reflects a much larger shift in oral science. Historically, dentistry was primarily mechanical: remove decay, rebuild structure, replace what has been lost. Those tools remain essential, but modern research increasingly asks whether molecular signaling can help explain why some tissues regenerate more effectively than others.
This has led to a growing focus on what may be called biomolecular dentistry or regenerative dentistry. In this framework, healing is not just a matter of materials and procedures. It is a matter of cellular instruction, biological timing, immune coordination, and tissue communication.
Areas of active interest include:
- periodontal ligament regeneration
- oral stem cell biology
- host-defense peptide function
- bone tissue engineering
- biomimetic enamel repair
- implant surface bioengineering
- connective tissue remodeling
As research develops, peptides may continue serving as valuable tools for understanding how tissues signal, repair, and reorganize. Whether future clinical applications emerge from these pathways remains a matter for ongoing scientific study, but the direction of interest is unmistakably molecular.
Frequently Asked Questions
What peptides are being researched for dental regeneration?
Researchers have explored molecules such as BPC-157, GHK-Cu, LL-37, TB-500, P11-4, and various growth or signaling proteins in discussions related to oral tissue repair, antimicrobial defense, enamel remineralization, and implant healing biology.
Can peptides help with dental implant healing?
Scientific interest in this area focuses on biological pathways involved in angiogenesis, connective tissue remodeling, immune balance, cell migration, and bone signaling. These are all important components of dental implant integration and healing research.
What is the P11-4 enamel peptide?
P11-4 is a biomimetic self-assembling peptide studied in dental research for its relationship to hydroxyapatite formation and early enamel remineralization models.
Why is LL-37 important in oral health research?
LL-37 is a host-defense peptide involved in innate immune function. In oral biology, it has been studied in relation to microbial regulation, biofilm interactions, mucosal defense, and inflammatory signaling.
What is regenerative dentistry?
Regenerative dentistry is an emerging research field focused on understanding and potentially supporting the body’s own repair systems in oral tissues such as gum, bone, enamel, and periodontal structures.
Conclusion
Peptides for dental regeneration remain an emerging but highly intriguing area of scientific research. From vascular and connective tissue signaling to antimicrobial defense and enamel biomimetics, current literature reflects growing interest in how molecular communication shapes oral healing.
What makes this field especially compelling is that it reframes dentistry through a biological lens. Rather than asking only how damaged tissue can be replaced, regenerative research asks how healing itself is organized, and whether that organization can be better understood through signaling molecules such as peptides.
For researchers interested in gum support, implant healing, oral tissue repair, and the future of biomolecular dentistry, peptides represent a valuable window into the body’s own regenerative language.
References
- Keremi B, Lohinai Z, Komora P, Duhaj S, Szekely AD, Jobbagy-Tenke P, et al. Antiinflammatory effect of BPC 157 on experimental periodontitis in rats. J Periodontal Res. 2009;44(6):717-725. PubMed
- Seiwerth S, Rucman R, Turkovic B, Sever M, Klicek R, Radic B, et al. Stable gastric pentadecapeptide BPC 157 and wound healing. Curr Pharm Des. 2021;27(14):1612-1632. PubMed
- Pickart L, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2018;19(7):1987. PubMed
- Tokajuk J, Deptuła P, Piktel E, Wollny T, Wolak P, Durnaś B, et al. Cathelicidin LL-37 in Health and Diseases of the Oral Cavity. Biomedicines. 2022;10(5):1086. PMC
- Goldstein AL, Hannappel E, Kleinman HK. Thymosin β4: a multi-functional regenerative peptide. Ann N Y Acad Sci. 2012;1270:1-6. PubMed
- Kirkham J, Firth A, Vernals D, Boden N, Robinson C, Shore RC, et al. Self-assembling peptide scaffolds promote enamel remineralization. J Dent Res. 2007;86(5):426-430. PubMed
- Díaz-Sánchez RM, Toro-Ibacache V, Mardones M, Penarrocha-Diago M. Current Approaches of Bone Morphogenetic Proteins in Dentistry. Int J Oral Maxillofac Surg. 2015;44(6):703-711. PubMed
- Nakashima M, Reddi AH. The application of bone morphogenetic proteins to dental tissue engineering. Nat Biotechnol. 2003;21(9):1025-1032. PubMed

Author Disclaimer:
The content published under this author’s byline is provided for informational and educational purposes only and reflects theoretical research discussions related to peptides, biochemistry, and related scientific topics.
Any credentials or academic titles referenced are academic in nature only and do not imply medical licensure, clinical authority, or the practice of medicine.
This content does not constitute medical advice, diagnosis, or treatment, and should not be interpreted as such. The author is not providing clinical guidance and is not acting as a healthcare provider.
All products discussed by BioGenix Peptides LLC are intended strictly for research, laboratory, and analytical use only and are not for human or animal consumption.
BioGenix Peptides LLC makes no claims regarding the safety, efficacy, or approved use of any compounds discussed.
Best selling products
-
Reta (GLP-3) 10mg
$120.00 -
Bacteriostatic Water (Reconstitution Solution) 10mL
$16.00 -
Reta (GLP-3) 20mg
$195.00 -
Bacteriostatic Water (Reconstitution Solution) 3mL
$9.00 -
BPC-157 + TB-500 20mg Blend
$105.00