LL-37 (Human Cathelicidin): A Multifunctional Host-Defense Peptide at the Center of Innate Immune Research
Research Use Only Disclaimer
This article is provided for informational and educational purposes only and reflects theoretical, research-focused discussions related to peptides, immunology, and biochemical signaling pathways. It does not constitute medical advice, diagnosis, or treatment. All compounds referenced are intended strictly for research, laboratory, and analytical use only and are not for human or animal consumption.
Introduction
LL-37 is frequently described as a human antimicrobial peptide, but modern research has revealed that this characterization captures only a fraction of its biological relevance. In contemporary literature, LL-37 is more accurately described as a multifunctional host-defense peptide with roles that extend well beyond direct antimicrobial activity.
Across immunology, dermatology, wound-environment research, and innate immune signaling studies, LL-37 has emerged as a molecule capable of interacting with microbial membranes, host cell receptors, extracellular nucleic acids, and inflammatory signaling pathways. This versatility makes it both a powerful research tool and a peptide that demands careful experimental design.
Origin and Biosynthesis of LL-37
LL-37 is the only member of the cathelicidin family expressed in humans. It is encoded by the CAMP gene and initially synthesized as a larger precursor protein commonly referred to as hCAP18. This precursor is biologically inactive until it undergoes extracellular proteolytic processing.
Multiple studies have demonstrated that enzymes such as proteinase-3 and kallikrein-5 cleave hCAP18 to generate the active LL-37 peptide, particularly in skin, immune, and wound-associated microenvironments. This processing step is critical, as dysregulation of cleavage has been implicated in inflammatory skin models and altered immune responses.
Expression of the CAMP gene is also tightly regulated. A substantial body of literature links LL-37 expression to vitamin D–responsive pathways, especially in epithelial cells and immune cells. This regulatory axis has positioned LL-37 at the intersection of barrier integrity, innate immunity, and environmental signaling.
Structural Features and Biophysical Behavior
LL-37 is a cationic, amphipathic peptide composed of 37 amino acids. In membrane-like environments, it commonly adopts an α-helical structure. This structural configuration underlies much of its biological behavior.
The combination of positive charge and amphipathic topology allows LL-37 to associate with negatively charged surfaces, including bacterial membranes, lipopolysaccharides (LPS), extracellular DNA, and RNA. Unlike highly target-specific ligands, LL-37 displays a form of “promiscuous binding,” which explains both its versatility and its context-dependent effects.
Biophysical studies emphasize that LL-37 does not act through a single rigid mechanism. Instead, its interactions vary depending on concentration, ionic strength, surrounding proteins, and the molecular composition of the local environment.
Antimicrobial and Membrane-Active Properties
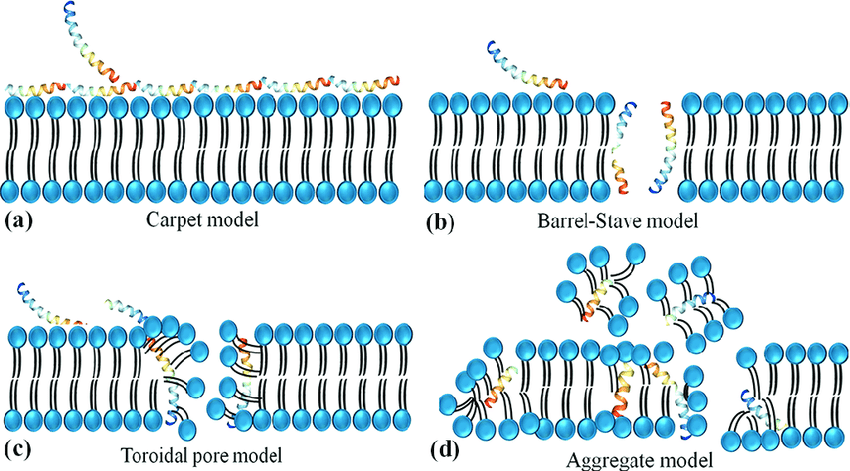
The earliest and most extensively studied role of LL-37 is its antimicrobial activity. In vitro models demonstrate that LL-37 can disrupt microbial membranes through mechanisms that include pore formation, membrane thinning, and destabilization of lipid bilayers.
Unlike classical antibiotics, LL-37 does not rely on a single molecular target. This broad membrane-active behavior has made it a frequent subject of interest in research exploring innate immune defenses against bacteria, fungi, and enveloped viruses.
However, researchers consistently note that antimicrobial efficacy is highly sensitive to experimental conditions. Salt concentration, serum proteins, and peptide concentration can dramatically alter observed outcomes, reinforcing the importance of context when interpreting LL-37 data.
Immunomodulatory and Chemotactic Signaling
Beyond direct antimicrobial effects, LL-37 functions as an immunomodulatory signaling molecule. Multiple studies describe its ability to influence leukocyte behavior, including chemotactic effects mediated through receptors such as formyl peptide receptor 2 (FPR2).
Through these interactions, LL-37 has been shown to participate in immune cell recruitment, cytokine modulation, and cross-talk between innate and adaptive immune responses in experimental systems. These signaling roles are central to why LL-37 appears in research spanning inflammation, tissue repair, and immune surveillance.
Importantly, LL-37’s immunological effects are not uniformly suppressive or activating. Depending on dose and environment, LL-37 may attenuate inflammatory signaling in some models while amplifying it in others.
LL-37 in Skin Biology and Barrier Research
Skin is one of the most extensively studied tissues for LL-37 activity. Keratinocytes, immune cells, and resident microbiota all contribute to a microenvironment where LL-37 expression and processing are tightly regulated.
In inflammatory skin research, LL-37 has been linked to disease-relevant signaling pathways. In rosacea-associated models, altered processing of cathelicidin into LL-37 fragments has been associated with exaggerated inflammatory responses. In psoriasis-related research, LL-37 has been implicated in pathways involving innate immune activation and nucleic-acid sensing.
These findings have shifted the perception of LL-37 from a purely protective peptide to a context-dependent immune modulator capable of contributing to pathological inflammation under specific conditions.
Interaction With Nucleic Acids and Innate Immune Amplification
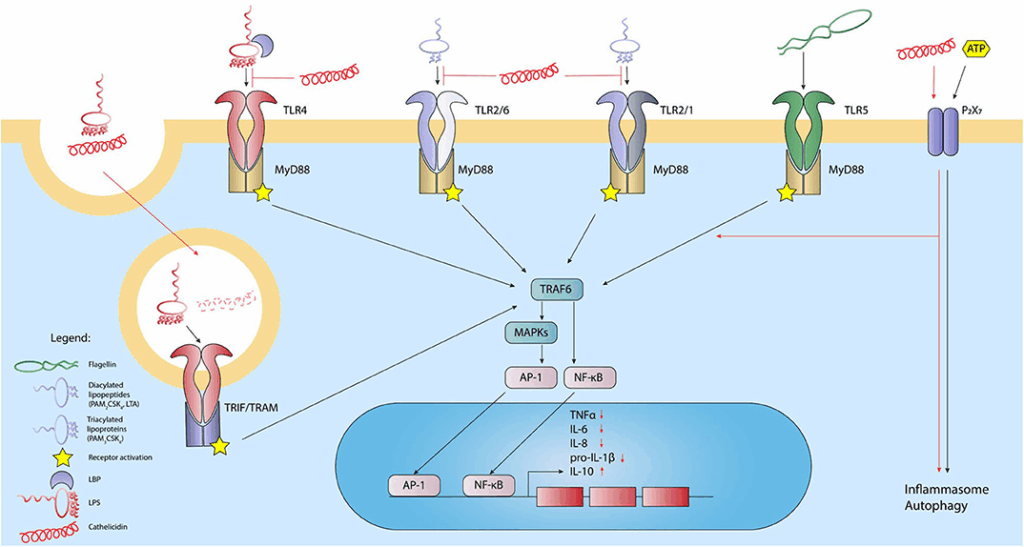
One of the most compelling areas of LL-37 research involves its interaction with extracellular DNA and RNA. LL-37 can form stable complexes with nucleic acids, altering how these molecules are internalized and sensed by immune cells.
In several experimental models, LL-37–nucleic acid complexes have been shown to enhance activation of innate immune receptors, including toll-like receptor pathways. This mechanism provides a potential explanation for LL-37’s role in autoimmune-like inflammation observed in certain skin and immune disorders.
From a research perspective, this property makes LL-37 a powerful tool for studying how endogenous peptides influence immune recognition of self-derived molecular patterns.
LL-37 in Wound-Environment and Tissue-Response Research
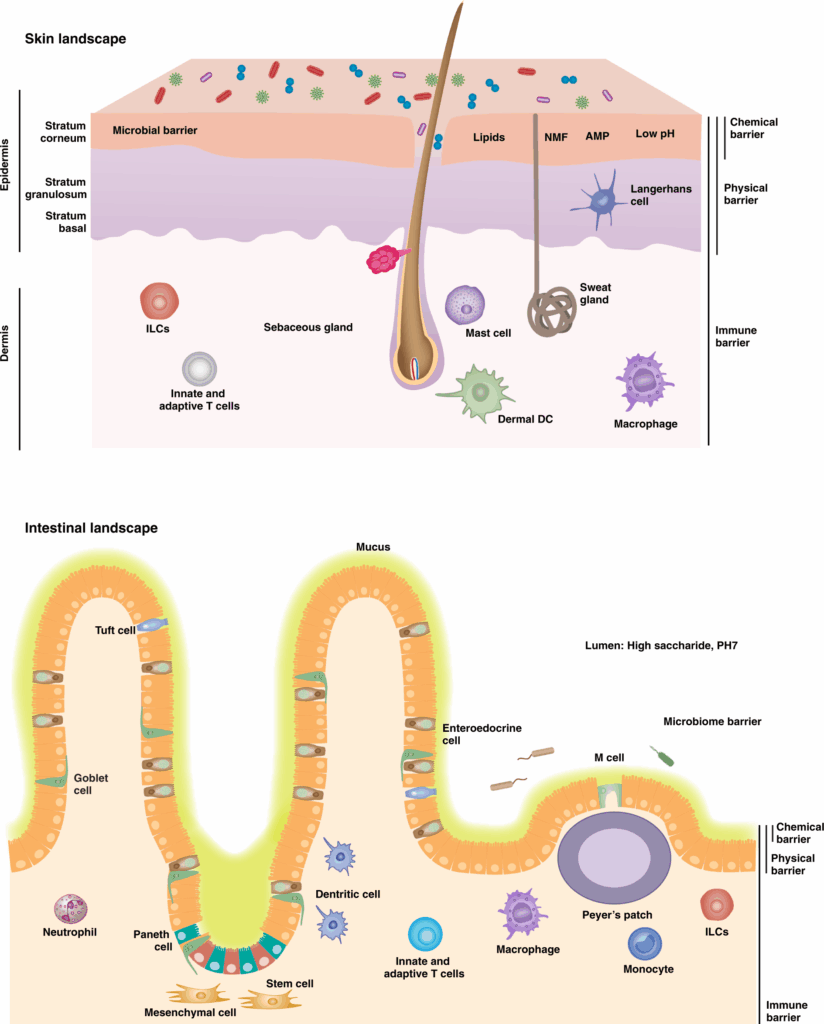
LL-37 is frequently studied in wound-environment models due to its ability to influence multiple phases of tissue response. Research literature describes roles for LL-37 in keratinocyte migration, re-epithelialization, angiogenic signaling, and modulation of inflammatory phases during wound repair.
Both preclinical and translational studies have explored LL-37-based systems to better understand how innate immune peptides coordinate microbial defense with tissue remodeling signals.
These multifaceted roles help explain why LL-37 is often discussed alongside other barrier- and tissue-focused research peptides in advanced experimental frameworks.
Endotoxin Binding and LPS Neutralization
Several studies have investigated LL-37’s ability to bind bacterial endotoxins such as lipopolysaccharide (LPS). In vitro and cell-based models suggest that LL-37 can neutralize LPS activity by direct binding, potentially reducing downstream inflammatory signaling in certain experimental conditions.
This property has positioned LL-37 as a molecule of interest in research focused on host responses to bacterial components rather than live pathogens alone.
Experimental Considerations and Research Cautions
The same features that make LL-37 attractive for research also introduce experimental complexity. Concentration-dependent effects are well documented, and supraphysiological levels may induce cytotoxic or pro-inflammatory outcomes not observed at lower concentrations.
Environmental factors—including ionic strength, serum presence, extracellular nucleic acids, and species-specific differences—can substantially alter LL-37 behavior. As a result, careful control selection and transparent reporting are essential when designing or interpreting LL-37 studies.
Rather than a limitation, this context sensitivity is precisely what makes LL-37 valuable for probing how innate immune systems balance defense, repair, and inflammation.
Related Research Peptides
LL-37 is frequently studied alongside other peptides involved in barrier integrity, immune signaling, and tissue-response research, including:
Peer-Reviewed References
- Kahlenberg JM, Kaplan MJ. Little peptide, big effects: the role of LL-37 in inflammation and autoimmune disease. J Immunol. 2013. PubMed
- Xhindoli D, et al. The human cathelicidin LL-37 — a pore-forming antimicrobial peptide and more. Biochim Biophys Acta. 2016. PubMed
- Sørensen OE, et al. Human cathelicidin, hCAP-18, is processed to the antimicrobial peptide LL-37 by proteinase-3. Blood. 2001. PubMed
- Heilborn JD, et al. The cathelicidin antimicrobial peptide LL-37 is involved in re-epithelialization of human skin wounds. J Invest Dermatol. 2003. PubMed
- Carretero M, et al. In vitro and in vivo wound healing-promoting activities of LL-37. J Invest Dermatol. 2008. PubMed
- Morizane S, et al. Cathelicidin antimicrobial peptide LL-37 in psoriasis. J Dermatol Sci. 2011. PubMed
- Takahashi T, et al. Cathelicidin promotes inflammation by enabling binding of self-RNA to Toll-like receptors. Nat Commun. 2018. PubMed
- Scott A, et al. Evaluation of the ability of LL-37 to neutralize lipopolysaccharide in vitro. J Endotoxin Res. 2011. PubMed
- Schauber J, et al. Injury enhances antimicrobial peptide expression through a vitamin D–dependent mechanism. J Clin Invest. 2007. PubMed
- Miao S, et al. Cathelicidin peptide LL-37: a multifunctional regulator of immune responses. Front Immunol. 2024. PubMed

LL-37 5mg
LL-37 is a naturally occurring peptide that plays an important role in the body’s first line of defense. It is part of the innate immune system and is studied for its ability to help protect the body from bacteria, support healthy inflammation balance, and assist in tissue repair.
In research settings, LL-37 has been shown to interact with harmful microbes, help maintain the strength of the skin and gut barriers, and support the body’s natural healing processes. Studies also explore its potential role in calming irritation, promoting wound recovery, and helping the immune system respond more effectively during stress or injury.
Because LL-37 can influence both immune activity and tissue repair, it is often used as a research tool to better understand how the body protects itself and restores damaged or irritated tissues.
In Stock




