How to Develop Robust HPLC Methods for Peptides
How to Develop Robust HPLC Methods for Peptides
Why Peptide Analysis by HPLC Is the Gold Standard for Research-Grade Characterization
Disclaimer: The information provided in this article is for educational and informational purposes only. It is not intended as medical advice, diagnosis, or treatment. Products and compounds referenced are for research use only and are not approved for human or veterinary consumption. Always consult a qualified professional regarding health or medical decisions.

Peptide analysis by HPLC is the most widely used method for measuring peptide purity, identifying impurities, and characterizing peptide structure in a research setting. Here is a quick overview of what it involves:
| What You Want to Know | Quick Answer |
|---|---|
| Best HPLC mode for peptides | Reversed-phase HPLC (RP-HPLC) with C18 or C4 columns |
| Typical mobile phase | Acetonitrile + 0.1% TFA in water, gradient elution |
| Detection wavelength | UV at 214-215 nm (peptide bond absorption) |
| Key purity metric | % main peak area by UV chromatogram |
| When to add MS | When impurity identification is needed |
| Column pore size for peptides | 300 Å for large peptides; 100 Å for small digest fragments |
Peptides are not simple molecules. They fold, they interact with surfaces, and two peptides that differ by a single amino acid can behave very differently on a column. That complexity is exactly why developing a robust HPLC method matters so much — a poorly designed method can miss critical impurities or give you unreliable purity data.
Over the past 25+ years, RP-HPLC has become the cornerstone analytical tool for peptide research, capable of separating peptides that differ by as little as one amino acid substitution and supporting sample loads from microgram-scale analytical work up to 200 mg in preparative applications.
This guide walks you through every key decision point — from column selection and mobile phase design to peptide mapping and regulatory-grade method validation — so you can build methods that are accurate, reproducible, and fit for purpose.
I’m Jay Daniel, Founder and CEO of BioGenix Peptides, with years of hands-on experience in peptide quality control, purity validation, and peptide analysis by HPLC across a wide range of research-grade compounds. In the sections below, I’ll break down the core principles and practical steps you need to develop methods that hold up under scrutiny.
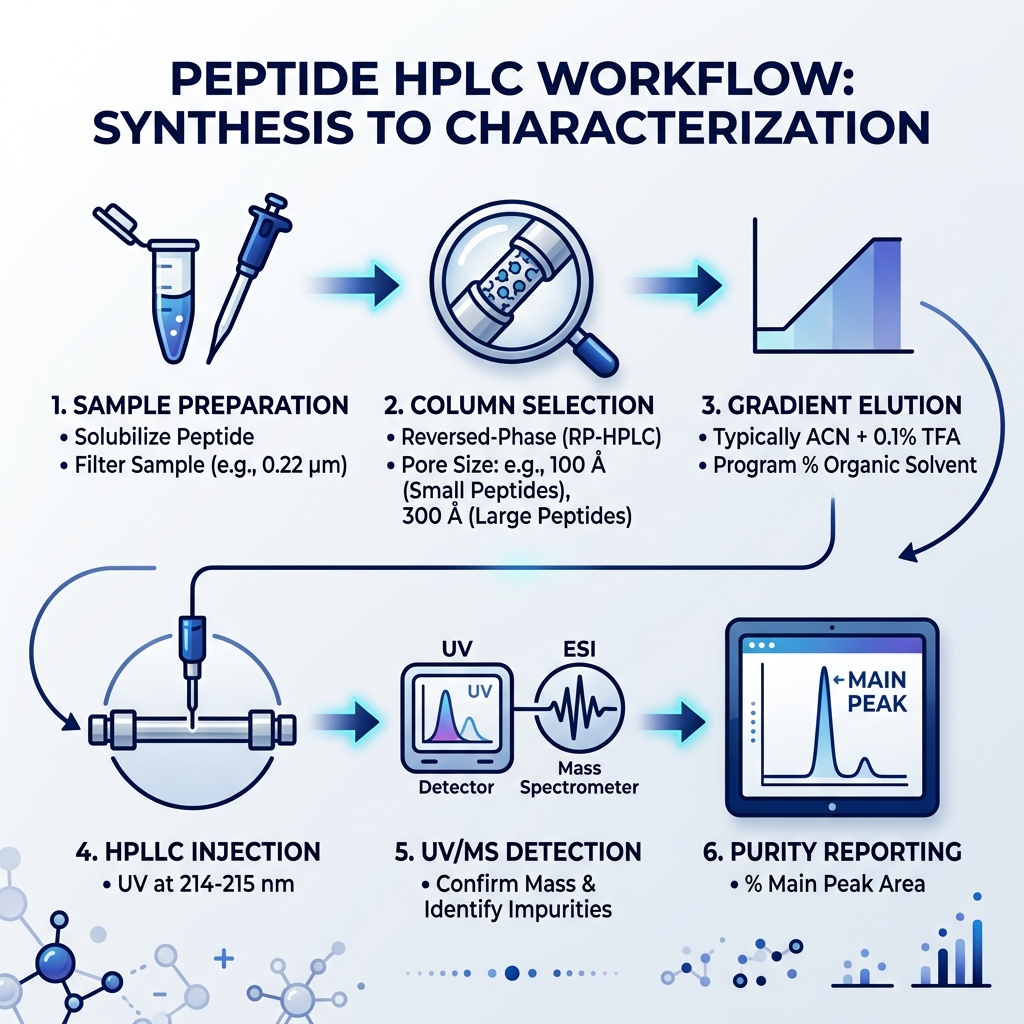
Core Chromatographic Modes and Mechanisms
When we talk about peptide analysis by hplc, we aren’t just looking at one specific technique. While one mode dominates the field, a truly robust research program often utilizes several “modes” to ensure no impurity goes unnoticed. Understanding how these molecules interact with the stationary phase is the first step in mastering the craft.
Primary Modes for Peptide Analysis by HPLC
There are four main “flavors” of HPLC used in peptide research:
- Reversed-Phase HPLC (RP-HPLC): This is the heavyweight champion. It separates peptides based on their hydrophobicity. Because most peptides have a mix of hydrophobic and hydrophilic amino acids, RP-HPLC offers incredible resolution. It is especially useful because the mobile phases (like water and acetonitrile) are volatile, making it easy to recover the peptide or link the system to a mass spectrometer.
- Size-Exclusion Chromatography (SEC): We use SEC primarily to look at the “size” or oligomerization state of a peptide. If a peptide is forming dimers or aggregates, SEC will show them as separate peaks. It’s a great way to ensure your research compound hasn’t started clumping together.
- Ion-Exchange Chromatography (IEX): This mode separates peptides based on their net charge. We often recommend strong cation-exchange (SCX) for general peptide work because peptides are highly soluble at low pH where their acidic groups are protonated.
- HILIC-CEX (Mixed-Mode): A newer approach that combines hydrophilic interaction chromatography with cation exchange. It can sometimes outperform RP-HPLC for very complex mixtures by separating peptides based on both charge and hydrophilicity.
For those interested in the chemistry behind these compounds, you can find more info about peptide synthesis to understand how the manufacturing process influences which HPLC mode is best for analysis.
Retention Mechanisms in Reversed-Phase Systems
The way a peptide sticks to an RP-HPLC column is fascinatingly different from small molecules. Small molecules tend to “partition” into the stationary phase, but peptides follow a “hydrophobic foot” mechanism.
Essentially, a peptide “adsorbs” onto the surface of the silica. It sits there until the concentration of the organic solvent (like acetonitrile) reaches a very specific point. At that precise moment, the peptide “desorbs” and zips through the column. This is why gradient elution is so critical for peptides; a tiny change in solvent concentration can be the difference between a peptide staying stuck and eluting perfectly.
The retention is driven by the amino acid side-chains and the peptide’s 3D folding. If a peptide is folded into an alpha-helix, it might hide some of its hydrophobic parts, changing how it interacts with the column. This is why we pay so much attention to temperature and pH—they can actually change the shape of the peptide while it’s inside the machine! To dive deeper into the technical protocols, you can review this Scientific research on HPLC analysis and purification.
Optimizing Mobile Phases and Column Parameters
Developing a robust method for peptide analysis by hplc requires a “Goldilocks” approach—everything from the acidity of the water to the size of the pores in the silica needs to be “just right.”
Mobile Phase Optimization for Peptide Analysis by HPLC
The mobile phase is the liquid that carries your sample through the column. For peptides, we almost always use a binary gradient system:
- Mobile Phase A: Water with an ion-pairing reagent (usually 0.1% TFA).
- Mobile Phase B: Acetonitrile with 0.1% TFA.
Why TFA? Trifluoroacetic acid (TFA) is an ion-pairing reagent. It does two things: it keeps the pH low (around 2.0), which suppresses the ionization of silanol groups on the column (reducing peak tailing), and it “pairs” with the positive charges on the peptide to make it more hydrophobic. Interestingly, research suggests that for very complex separations, increasing TFA to 0.2% or 0.25% can actually improve resolution even further.
Gradient Slope: Unlike small molecules where you might jump from 5% to 95% solvent quickly, peptides require shallow gradients. A typical starting point is a 1% increase in organic solvent per minute. For extremely similar variants, we might go as low as 0.05% to 0.25% per minute.
If you are reviewing a lab report, knowing more info about reading a peptide COA can help you understand how these mobile phase choices affect the final purity numbers.
Column Characteristics for Enhanced Resolution
Choosing the right column is half the battle. Here are the critical factors:
- Pore Diameter: This is the most common mistake in peptide HPLC. Standard columns have 100Å (Angstrom) pores, which are fine for small peptides. However, larger peptides and proteins can’t fit into those tiny holes. For these, we use 300Å wide-pore silica. If the peptide can’t enter the pore, it can’t interact with the stationary phase, leading to poor resolution.
- Bonded Phases (C18 vs. C4):
- C18: The standard for small to medium peptides (<3,000 Da). It provides the most "grip."
- C4 or C8: Better for very hydrophobic peptides or large proteins. A C18 might hold onto a hydrophobic protein so tightly it never comes off!
- Column Temperature: We often run peptide HPLC at elevated temperatures (40°C to 60°C, and sometimes up to 90°C). Heat reduces the viscosity of the mobile phase, leading to sharper peaks and faster separations. It can even change the selectivity, helping to resolve two peaks that overlap at room temperature.
| Feature | Small Peptides (<20 AA) | Large Peptides/Proteins |
|---|---|---|
| Pore Size | 100 – 120 Å | 300 Å |
| Stationary Phase | C18 | C4 or C8 |
| Sensitivity Focus | Standard 4.6mm ID | Narrowbore (2.1mm) |
Advanced Characterization: Mapping and MS Integration
Sometimes, simply knowing a peptide is “98% pure” isn’t enough for high-level research. We need to know what that other 2% is, or we need to confirm the exact sequence. This is where advanced techniques like peptide mapping and Mass Spectrometry (MS) come into play.
Peptide Mapping and Structural Fingerprinting
Peptide mapping is like taking a fingerprint of a protein. We take a large protein or long-chain peptide and “cut” it into smaller pieces using an enzyme—most commonly trypsin. Trypsin is very predictable; it cuts the chain at specific amino acids (Lysine and Arginine).
On average, trypsin digestion produces one peptide for every 10-12 amino acids. By running these fragments through an RP-HPLC, we create a “map.” If a single amino acid in the original protein was oxidized or deamidated, the peak for that specific fragment will shift. For example, a methionine oxidation will cause a peptide to elute earlier because it becomes more hydrophilic.
This level of detail is critical for identifying post-translational modifications (PTMs) and ensuring batch-to-batch consistency. You can see how experts handle these complex datasets in this Scientific research on peptide characterization.
Coupling HPLC with Mass Spectrometry
While UV detection (at 214 nm) tells us how much of something is there, Mass Spectrometry tells us what it is. Coupling the two (LC-MS) is the ultimate diagnostic tool for peptide analysis by hplc.
However, there is a catch: TFA, our favorite ion-pairing reagent, is a “signal killer” in mass spectrometry. It suppresses the ionization process. To fix this, researchers often:
- Reduce TFA concentration to 0.01% or lower.
- Switch to Formic Acid or Acetic Acid (though this can hurt chromatographic resolution).
- Use “TFA Fix” solutions or specialized column chemistries that work well with low-acid mobile phases.
For a deeper dive into how these two technologies work together, check out our guide on HPLC and Mass Spectrometry in Peptide Analysis.
Regulatory Standards for Peptide Analysis by HPLC
In pharmaceutical development (NDAs and ANDAs), HPLC is more than a research tool—it’s a regulatory requirement. Regulators expect a “complete” picture of the peptide’s purity profile.
Purity Assessment and Related Substances
When we analyze a peptide for a regulatory submission, we aren’t just looking at the main peak. We are hunting for “related substances.” These include:
- Truncated Sequences: Peptides that are missing one or more amino acids.
- Deamidated or Oxidized Species: Chemical “damage” to the peptide.
- Diastereomers: Peptides with the same sequence but different 3D orientations (D-amino acids instead of L-amino acids).
Regulators generally expect identification of any impurity that appears consistently above a certain threshold (often 0.1%). This requires high-resolution methods that can distinguish between the main component and these very similar “neighbors.”
Method Validation for Research and Development
A robust method must be validated for:
- Selectivity: Can the method actually separate the peptide from its known impurities?
- Linearity: Does the peak area increase proportionally with the amount of sample?
- Robustness: If the temperature drifts by 2 degrees or the pH changes by 0.1, does the method still work?
For researchers, staying updated on HPLC-MS categories is a great way to ensure your analytical strategies align with current industry standards.
Frequently Asked Questions about Peptide HPLC
What is the best pore size for peptide analysis?
For most synthetic peptides under 3,000 Daltons, a 100Å to 120Å pore size is sufficient. However, for larger peptides, long-chain analogs, or proteins, 300Å is the industry standard. Using a pore size that is too small leads to “restricted diffusion,” which results in broad, ugly peaks and poor recovery.
How does temperature affect peptide resolution?
Temperature is a powerful tool in peptide analysis by hplc. Increasing the temperature (typically to 40-60°C) lowers the mobile phase viscosity and improves mass transfer. This usually results in narrower peaks and better resolution. More importantly, temperature can change the peptide’s conformation. Occasionally, two peptides that co-elute at 25°C will perfectly separate at 50°C because their shapes change differently with heat.
Why is TFA used in peptide mobile phases?
TFA (Trifluoroacetic acid) serves two roles. First, it acts as a buffer to keep the pH low, which prevents the peptide from interacting with “active” sites on the silica (silanols) that cause peak tailing. Second, it acts as an ion-pairing agent. It “wraps” around the positively charged parts of the peptide, making it more hydrophobic and helping it stick to the column. Without TFA, many peptides would simply wash through the column without any separation.
Conclusion
Developing robust methods for peptide analysis by hplc is both a science and an art. It requires a deep understanding of how amino acids behave under pressure, heat, and varying chemical environments. Whether you are performing a simple purity check or a complex tryptic map, the choices you make regarding column chemistry, mobile phase composition, and temperature will define the quality of your research data.
At BioGenix Peptides, we pride ourselves on analytical excellence. We understand that in research, the integrity of your compound is everything. By utilizing state-of-the-art RP-HPLC and LC-MS techniques, we ensure that every peptide meets the rigorous standards required for high-level scientific inquiry.
If you’re looking to improve your lab’s internal processes, consistency is key. Always use high-purity reagents, maintain your columns, and never underestimate the power of a shallow gradient. To ensure your compounds remain in peak condition after analysis, be sure to Learn more about peptide storage and stability for the best practices in maintaining research-grade integrity.
By following the systematic approach outlined in this guide, you can develop HPLC methods that are not only robust but also capable of uncovering the fine details of peptide structure and purity that others might miss.



