Research Peptides: Terms Demystified
What Are Research Peptides? A Plain-English Overview
Research peptides are short chains of amino acids synthesized in a laboratory setting and sold strictly for scientific, in-vitro, and analytical purposes — not for human or animal use.
Quick answer:
| Question | Answer |
|---|---|
| What are they? | Synthetic amino acid chains used in laboratory research |
| Are they for human use? | No — strictly for in-vitro lab research only |
| Are they FDA-approved drugs? | No — not evaluated by the FDA as medicines |
| Who can use them? | Qualified, trained research professionals |
| Where are they sold? | Specialist suppliers, not pharmacies |
The peptide field has exploded in recent years. Over 11% of all new pharmaceutical chemical entities approved by the FDA between 2016 and 2024 were synthetic peptides. The broader peptide therapeutics market generated over $70 billion in global sales in 2019 — and it’s still growing fast.
But here’s where many people get confused.
The peptides driving that growth go through years of clinical testing, strict manufacturing controls, and full regulatory review before reaching patients. Research peptides occupy a completely different category. They are tools for scientific discovery — not finished medicines.
Yet the terminology around them is often murky. Terms like “in-vitro use only,” “not for human consumption,” and “research grade” get thrown around without much explanation. That leaves a lot of researchers — and curious readers — unsure of what they’re actually dealing with.
This guide cuts through that confusion.
I’m Jay Daniel, Founder and CEO of BioGenix Peptides, and I’ve spent years working directly in the research peptides industry — overseeing quality control, sourcing protocols, and manufacturing standards for laboratory-grade peptide products. My goal here is to give you a clear, honest breakdown of the terms, rules, and science behind research peptides so you can work with confidence.
The Regulatory Framework Governing Research Peptides
When we talk about research peptides, we aren’t talking about the insulin or GLP-1 agonists you find at a local pharmacy. Those are therapeutic peptides. Research peptides exist in a specific legal “carve-out” intended to facilitate scientific advancement without the multi-billion dollar overhead of clinical drug approval.
In the United States, the FDA maintains strict oversight on what constitutes a “drug.” According to FDA guidance on synthetic peptides, any substance intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease is a drug. Research peptides are explicitly not intended for these purposes.
To stay compliant, reputable suppliers must adhere to several key distinctions:
- Non-Therapeutic Status: These chemicals are not evaluated by the FDA for safety or efficacy in humans.
- 503A/503B Distinction: Research peptide suppliers are not compounding pharmacies (503A) or outsourcing facilities (503B). They do not fill prescriptions for individual patients.
- Legal Disclaimers: Every vial is typically labeled with “Not for Human Consumption” or “For Research Purposes Only.”
Understanding these Peptide Basics is crucial for any lab manager or scientist. The regulatory environment ensures that while these compounds are accessible for study, they remain separated from the consumer medical market.
Why Research Peptides are Labeled for In-Vitro Use Only
You’ve likely seen the bold warning: “In-Vitro Laboratory Use Only.” This isn’t just a suggestion; it’s a critical legal and safety boundary.
“In-vitro” refers to studies performed outside of a living organism, such as in a petri dish or test tube. Here is why research peptides carry this label:
- Safety Hazards: Many synthetic peptides have unknown toxicity profiles in living systems. Handling them requires professional laboratory equipment and PPE.
- Non-Sterilized: Unlike clinical-grade injectables, research peptides are often not manufactured in a sterile environment intended for human entry. They may contain trace amounts of solvents or salts that are harmless in a test tube but dangerous in a bloodstream.
- TSCA Inventory: Most of these chemicals are not on the Toxic Substances Control Act (TSCA) inventory because they are produced in small quantities for R&D.
- Analytical Purposes: They are designed for calibration, mass spectrometry, or EMA immunogenicity assessment models, not for metabolism by a biological liver or kidney.
Identifying Quality Suppliers of Research Peptides
In a market that can sometimes feel like the “Wild West,” finding a trustworthy source is paramount. We always advocate for transparency. A high-quality supplier should provide:
- Purity Standards: Look for a minimum of 98% purity, verified by third-party testing.
- USA Manufacturing: Peptides synthesized in the USA often adhere to more rigorous documentation and quality control standards.
- Technical Support: Can the company explain the salt content of their product? Do they offer The Complete Guide to Peptides in Scientific Research?
- Custom Synthesis: The ability to create specific sequences (custom synthesis) shows a higher level of chemistry expertise than a simple reseller.

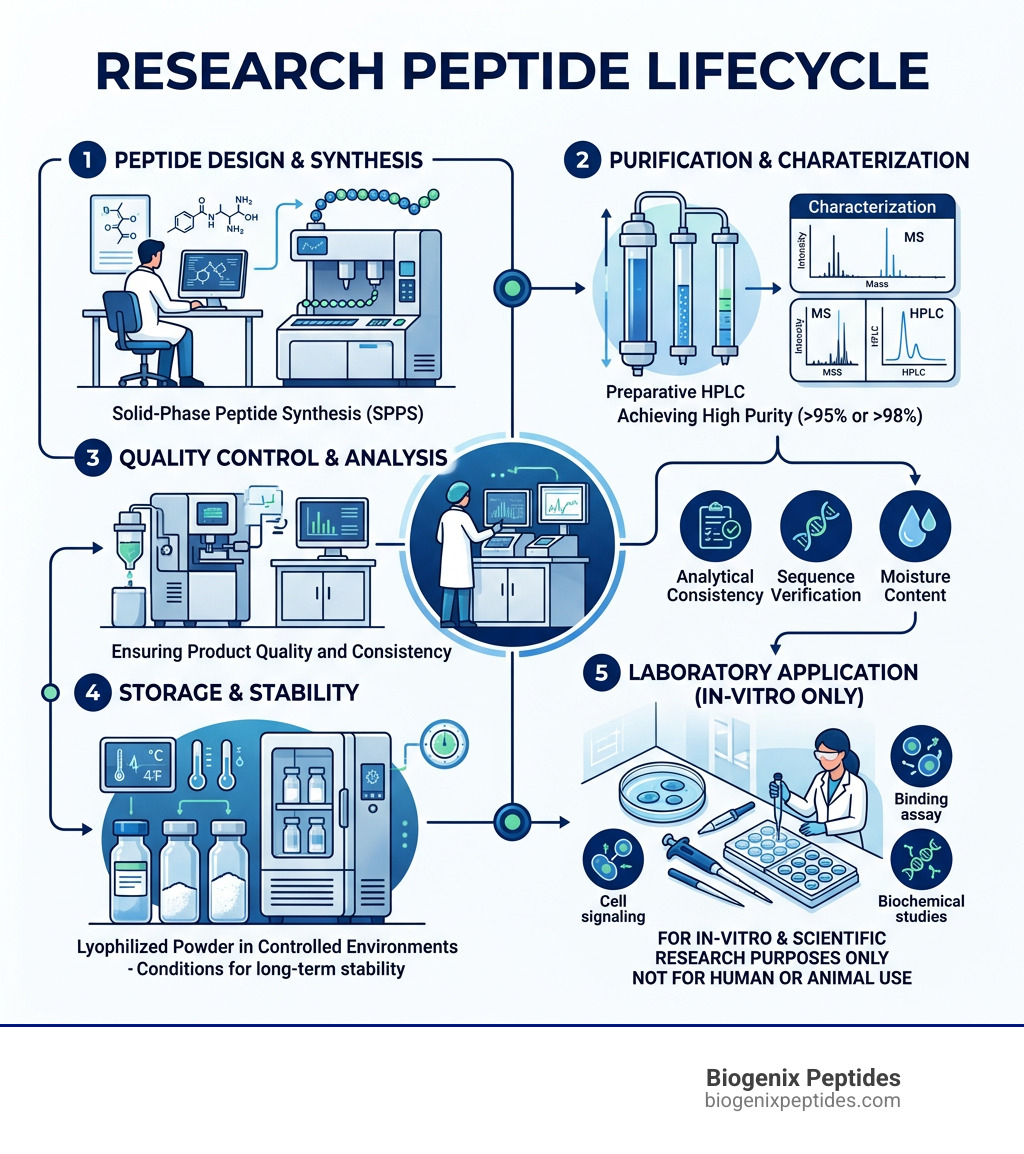
The Science of Synthesis: Manufacturing and Impurities
Creating research peptides is a feat of molecular engineering. There are two primary methods used: Solid Phase Peptide Synthesis (SPPS) and Liquid Phase Peptide Synthesis (LPPS).
Most modern research labs utilize SPPS, a method where the peptide chain is built on a solid resin. This allows for rapid production and easier washing of the growing chain. However, this process is notoriously “dirty” from an environmental perspective. It is estimated that peptide manufacturing produces 3 to 15 tons of waste per kg of peptide yield.
Recently, there has been a significant shift toward Green Chemistry. On December 12, 2023, the EU restricted the use of DMF (Dimethylformamide), a common but toxic solvent used in SPPS. This move is forcing the industry to adopt greener alternatives like EtOAc or DMSO, which may impact the types of impurities we see in the final product.
| Feature | Solid Phase (SPPS) | Liquid Phase (LPPS) |
|---|---|---|
| Speed | Very Fast | Slower |
| Scalability | Good for small/medium batches | Excellent for large-scale |
| Purity | High, but requires extensive washing | Can be easier to purify mid-process |
| Waste | High solvent usage | Generally lower waste |
To learn more about the technical nuances, check out our deep dive: Peptide Synthesis Explained: Solid Phase vs Solution Phase.
Managing Impurities in Synthetic Peptides
No chemical synthesis is 100% perfect. During the building of a peptide, “deletions” (where an amino acid is skipped) or “truncations” (where the chain stops growing) can occur.
For therapeutic approval (ANDA), the FDA is incredibly strict:
- Impurity levels must be equal to or less than the Reference Listed Drug (RLD).
- Any new impurity greater than 0.5% is generally unacceptable.
- Impurities between 0.1% and 0.5% must be identified and characterized.
In research peptides, managing these Scientific research on peptide impurities is the difference between a successful experiment and a skewed result. If a peptide is 95% pure, that 5% of “other stuff” could potentially interfere with cellular signaling in your assay.
Assessing Immunogenicity and Safety in Research Peptides
One of the hottest topics in peptide science right now is immunogenicity—the ability of a substance to provoke an immune response. Even if a peptide sequence is “natural,” synthetic versions can cause the body to create Anti-Drug Antibodies (ADAs).
Researchers use a “tiered” approach to assess this risk:
- In Silico: Using computer algorithms to predict if a peptide sequence will bind to HLA (Human Leukocyte Antigen) alleles.
- In Vitro: Testing the peptide against human PBMCs (Peripheral Blood Mononuclear Cells) to see if T-cells activate.
- In Vivo: Using transgenic or “humanized” mice that possess human immune components.
Understanding this risk is vital for Research on immunogenicity risk because impurities—even at very low levels—can act as “adjuvants,” essentially “tricking” the immune system into attacking the peptide. This is why learning How to Read a Peptide COA: Understanding HPLC & Mass Spectrometry (MS) is a non-negotiable skill for modern researchers.
Advanced Assays for Peptide Evaluation
To get a more granular look at safety, scientists use MAPPs assays (MHC-Associated Peptide Proteomics). This involves:
- Exposing Dendritic Cells (DCs) to the peptide.
- Allowing the cells to “process” and present the peptide fragments on their surface.
- Using mass spectrometry to see exactly which fragments the immune system “sees.”
These Clinical trial data on immunogenicity help researchers predict how a peptide might behave in a complex biological environment before they ever move to animal models.
Market Evolution and Future Trends in Peptide Research
The future of research peptides is bright, driven by massive investment in biotechnology. Between 2016 and 2024, 48 out of 420 new drug approvals (over 11%) were synthetic peptides. In 2023 alone, 16% of total FDA approvals were peptides.
We are seeing a massive shift toward:
- Bioregulators: These are ultra-short peptides (2–4 amino acids) that act as epigenetic switches. They are The Ultra-Short Peptides Changing How We Think About Cellular Signaling.
- Life Cycle Assessment (LCA): As the industry grows, researchers are looking at the “cradle-to-grave” environmental impact of peptide production.
- Preclinical Pipeline: There are currently approximately 600 peptide therapeutics in preclinical development and another 200 in clinical testing.
This growth means that the demand for high-quality research peptides will only increase as scientists scramble to study these new sequences.
Frequently Asked Questions about Research Peptides
What is the difference between research and therapeutic peptides?
The primary difference is intended use and regulatory status. Therapeutic peptides are FDA-approved (or in the approval pipeline) for human use and must be manufactured under cGMP (current Good Manufacturing Practice) conditions. Research peptides are chemicals sold for laboratory study only. Using research peptides for human consumption is one of The 10 Most Common Mistakes People Make When They’re New to Peptide Research.
Why are research peptides strictly for laboratory use?
Because they haven’t been through the rigorous safety and toxicity testing required for human consumption. They may contain synthesis byproducts, residual solvents, or salts that are not safe for ingestion or injection. Furthermore, the legal framework allows these compounds to be sold specifically to advance science, not to bypass drug laws.
How should research peptides be stored for stability?
Most research peptides are sold as “lyophilized” (freeze-dried) powders. For long-term stability:
- Store at -20°C or -80°C for maximum shelf life.
- Avoid moisture: Keep vials tightly sealed.
- Reconstitute only when ready: Once a peptide is in a liquid solution, it begins to degrade much faster. For a step-by-step guide, see our Peptide Storage & Stability Guidelines.
Conclusion
At Biogenix Peptides, we believe that the next great medical breakthrough is currently sitting in a laboratory vial. Research peptides are the building blocks of that future. By understanding the regulatory framework, the science of synthesis, and the importance of purity, researchers can push the boundaries of what’s possible in cellular signaling and metabolism.
If you are ready to begin your next phase of discovery, we invite you to explore our catalog of high-purity compounds.


