How to Read a Peptide COA: Understanding HPLC & Mass Spectrometry (MS)
Research-Only Disclaimer:
This content is provided for informational and educational purposes only and reflects theoretical discussions related to analytical chemistry and peptide research. It does not constitute medical advice, diagnosis, or treatment. All products discussed by BioGenix Peptides LLC are intended strictly for research, laboratory, and analytical use only and are not for human or animal consumption.
What high-quality Certificates of Analysis actually tell you — and how to spot red flags.
Why COAs Matter (And Why “99%” Isn’t Enough)
In the peptide research space, Certificates of Analysis (COAs) are frequently referenced but rarely understood. Many suppliers display a purity percentage without explaining how that value was generated, which analytical methods were used, or what researchers should actually look for.
This guide explains the two analytical pillars that matter most on a legitimate COA: HPLC for purity assessment and Mass Spectrometry for molecular identity confirmation.
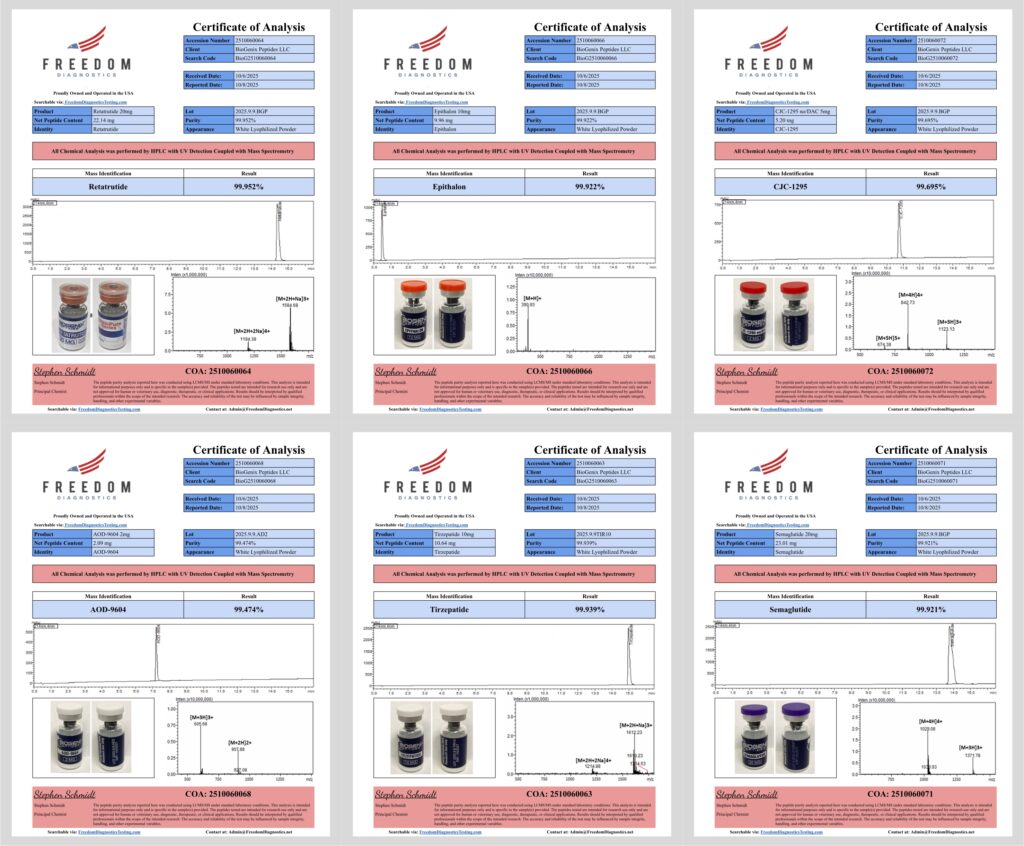
What Is a Certificate of Analysis (COA)?
A Certificate of Analysis is a laboratory-generated document reporting analytical test results for a specific compound and a specific production lot. A real COA ties results directly to the exact material being sold.

Example: BioGenix Peptides U.S., 3rd Party, Independent HPLC & MS Lab test for 20mg Retatrutide. (Freedom Diagnostics)
At minimum, a COA should include:
- Product name and identity
- Lot or batch number
- Analytical methods used (HPLC, MS, etc.)
- Purity results
- Test dates and laboratory information
If any of these elements are missing, the COA is incomplete.
HPLC: How Peptide Purity Is Measured
What is HPLC?
High-Performance Liquid Chromatography (HPLC) is the primary method used to estimate peptide purity. The sample is separated inside a chromatography column, and the components are detected as peaks on a graph called a chromatogram.
How to read an HPLC chromatogram
- One dominant peak: A clean peptide sample typically shows one large peak representing the target compound.
- Minimal secondary peaks: Smaller peaks may represent minor impurities or synthesis byproducts. Large secondary peaks are a warning sign.
- Retention time: The main peak appears at a consistent time under the same analytical method.
- Purity percentage: Purity is calculated as the area of the main peak relative to total detected peaks.
Important: HPLC purity is method-dependent. Columns, solvents, gradients, and detection wavelengths all influence results. That is why reputable COAs specify the analytical method used.
Mass Spectrometry (MS): Confirming Molecular Identity
What MS confirms
While HPLC estimates purity, Mass Spectrometry (MS) confirms identity. MS verifies that the compound’s molecular weight and charge states match the expected peptide structure.
What to look for on an MS spectrum
- Expected mass-to-charge (m/z) values
- Common peptide charge states (often +2 or +3)
- Major peaks aligning with theoretical molecular weight
MS helps confirm that the material is not just clean — it is the correct peptide.
Key COA Fields You Should Always Review
1. Product & Identity
Ensure the product name matches exactly and that identity is confirmed using MS or equivalent techniques.
2. Lot Number
Each production run should have a unique lot number. Reused COAs across multiple lots are a major red flag.
3. Net Peptide Content
Net peptide content reflects the peptide amount excluding salts or counter-ions and is important for analytical accuracy.
4. Purity & Method
Purity values should always be paired with a named analytical method such as HPLC-UV or LC-MS. Statements like “tested for purity” without method details lack transparency.
5. Appearance
Physical descriptors such as “white lyophilized powder” help verify consistency between lots.
6. Dates & Lab Information
Look for received, tested, and reported dates along with identifiable laboratory and analyst information.
Common COA Red Flags
- No lot or batch number
- No analytical method listed
- No chromatogram or MS data provided
- Generic “99% purity” claims without context
- COAs reused across multiple products
- Dates that do not align with the product being sold
Why BioGenix Emphasizes Transparency
At BioGenix Peptides LLC, Certificates of Analysis are treated as analytical documentation, not marketing material. Our COAs emphasize lot-specific reporting, named analytical methods, and clear laboratory data so researchers can evaluate results with confidence.
In Simple Terms
- HPLC tells you how clean it is
- MS tells you what it is
- A real COA should show both

Note: Labels shown on internal test samples may not reflect the exact appearance of the labels on distributed products.


