AMPK Activation & Mitochondrial Metabolic Control
How Cells Decide When to Burn, Build, or Conserve Energy
Research Use Only: This article is provided for educational and informational purposes only. It discusses theoretical and preclinical research related to cellular metabolism and biochemical signaling pathways. It does not constitute medical advice. All compounds and mechanisms referenced are discussed strictly within a research context and are not intended for human or animal use.
Introduction: Metabolism Is a Decision-Making System
Metabolism is often described as a simple balance between calories consumed and calories expended. In reality, metabolism is governed by intracellular decision-making systems that determine whether energy is stored, mobilized, conserved, or invested in repair and adaptation.
At the center of this decision-making process is AMP-activated protein kinase (AMPK), a highly conserved cellular energy sensor that links nutrient availability, mitochondrial output, and long-term metabolic efficiency.
Understanding AMPK activation is essential for understanding how cells regulate mitochondrial function, metabolic flexibility, and energetic resilience under stress.
What Is AMPK?
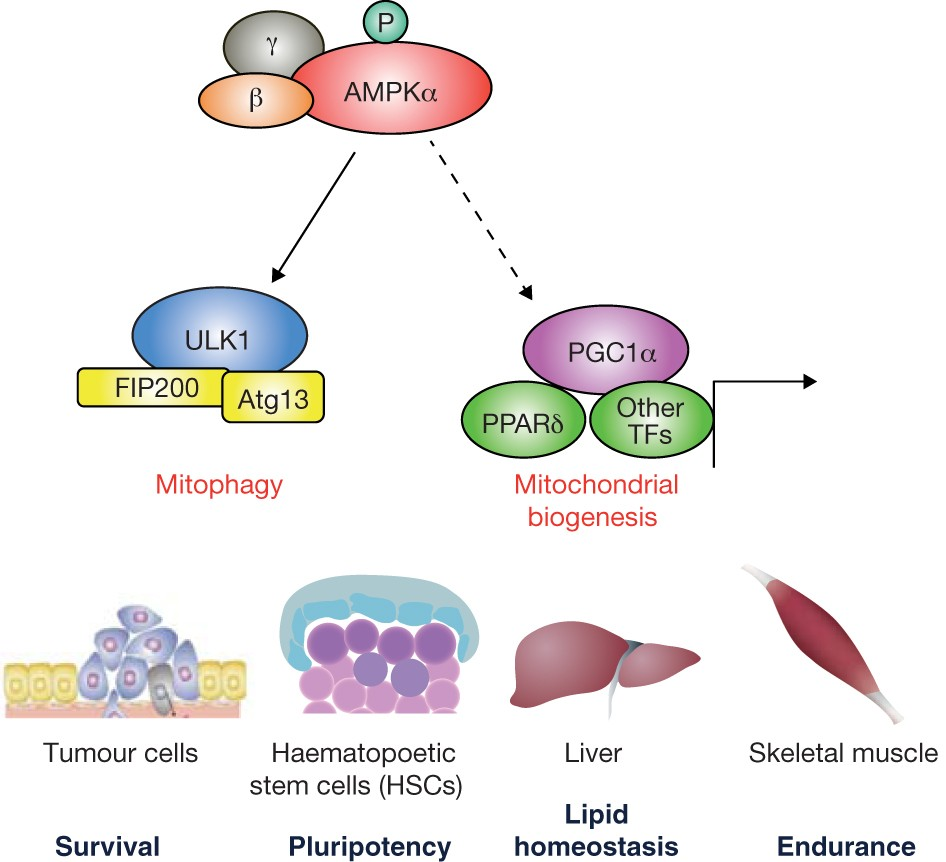
AMPK is an intracellular enzyme complex that functions as a metabolic “fuel gauge.” It becomes activated when cellular energy levels decline, reflected by an increased AMP-to-ATP ratio.
Once activated, AMPK initiates a coordinated shift in cellular behavior:
- Energy-consuming (anabolic) processes are suppressed
- Energy-generating (catabolic) pathways are enhanced
- Mitochondrial efficiency and substrate utilization are optimized
Rather than forcing immediate energy expenditure, AMPK improves how efficiently energy is produced and allocated.
AMPK as a Master Regulator of Mitochondrial Function
One of AMPK’s most critical roles is its influence over mitochondrial metabolism. Mitochondria are not static power plants — they dynamically adapt their number, efficiency, and fuel preference based on intracellular signaling cues.
AMPK activation affects mitochondria in several key ways:
- Enhances mitochondrial biogenesis through transcriptional coactivators
- Improves fatty acid transport and oxidation
- Reduces oxidative stress by improving electron transport efficiency
- Promotes removal of dysfunctional mitochondria via mitophagy
These changes do not increase energy output indiscriminately. Instead, they increase metabolic efficiency — more usable energy from fewer resources.
Metabolic Flexibility and Fuel Switching
Metabolic flexibility refers to a cell’s ability to switch between substrates such as glucose, fatty acids, and ketone bodies based on availability and demand.
AMPK plays a central role in this flexibility by:
- Suppressing glucose storage pathways during energy stress
- Upregulating fatty acid oxidation enzymes
- Reducing lipid accumulation within non-adipose tissues
In research settings, impaired AMPK signaling is consistently associated with mitochondrial inefficiency, ectopic fat accumulation, and metabolic inflexibility.
AMPK, Mitochondrial Quality Control, and Longevity Signaling
Beyond acute energy regulation, AMPK is deeply involved in long-term cellular maintenance. It coordinates with autophagy and mitophagy pathways to remove damaged proteins and dysfunctional mitochondria.
This quality-control role is particularly important because dysfunctional mitochondria generate excess reactive oxygen species and inefficient ATP output.
By promoting mitochondrial turnover and renewal, AMPK signaling contributes to:
- Improved cellular resilience under stress
- Reduced inflammatory signaling
- Preservation of metabolic efficiency over time
This has made AMPK a central focus in aging, metabolic disease, and mitochondrial research.
AMPK Is Not a “Fat-Burning Switch”
A common misconception is that AMPK activation simply increases fat burning. In reality, AMPK does not prioritize weight loss or fat reduction.
Its priority is survival efficiency.
AMPK activation reallocates resources away from growth and storage toward repair, maintenance, and energy balance. Any changes in body composition observed in research contexts are downstream effects of improved metabolic control — not the primary objective of the pathway itself.
Why AMPK Is Central to Modern Metabolic Research
AMPK sits at the intersection of nutrient sensing, mitochondrial signaling, and cellular adaptation. It integrates inputs from:
- Energy availability
- Oxidative stress
- Inflammatory signaling
- Mitochondrial performance
Because of this central role, AMPK is increasingly studied not as a target for acute stimulation, but as a gateway into understanding how metabolic systems adapt over time.
Modern research has shifted from asking “how much energy is burned” to asking “how efficiently energy systems are regulated.” AMPK is foundational to that shift.
Conclusion: Efficiency Over Intensity
AMPK activation represents a cellular philosophy of efficiency rather than excess. It does not force metabolism into overdrive — it refines, optimizes, and preserves.
By coordinating mitochondrial function, substrate selection, and cellular maintenance, AMPK ensures that energy systems remain adaptable under both abundance and stress.
As metabolic research continues to evolve, AMPK remains one of the most critical frameworks for understanding mitochondrial control, metabolic health, and long-term cellular resilience.
Peer-Reviewed References
- Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012;13(4):251–262. https://www.nature.com/articles/nrm3311
- Garcia D, Shaw RJ. AMPK: mechanisms of cellular energy sensing and restoration of metabolic balance. Mol Cell. 2017;66(6):789–800. https://www.sciencedirect.com/science/article/pii/S1097276517302917
- Herzig S, Shaw RJ. AMPK: guardian of metabolism and mitochondrial homeostasis. Nat Rev Mol Cell Biol. 2018;19(2):121–135. https://www.nature.com/articles/nrm.2017.95
- Canto C, Auwerx J. AMPK signaling and its downstream effects on mitochondrial biogenesis. Cell Metab. 2010;11(4):255–267. https://www.sciencedirect.com/science/article/pii/S1550413110000782
- Mihaylova MM, Shaw RJ. The AMPK signalling pathway coordinates cell growth, autophagy, and metabolism. Nat Cell Biol. 2011;13(9):1016–1023. https://www.nature.com/articles/ncb2329


